Product Description
Vamorolone is a first-in-class drug candidate that binds to the same receptors as corticosteroids but modifies the downstream activity of the receptors1,2. This has the potential to 'dissociate' efficacy from typical steroid safety concerns and therefore could emerge as a valuable alternative to corticosteroids, the current standard of care in children and adolescent patients with DMD. There is a clear unmet medical need in this patient group as high dose corticosteroids have significant systemic side effects that detract from patient quality of life. On September 2, 2020, Santhera exercised its option and obtained worldwide rights to vamorolone in Duchenne muscular dystrophy and all other indications. (Sourced from: https://www.santhera.com/health-care-professionals/vamorolone)
Mechanisms of Action: Steroid receptor Agonist
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: Priority Review - Muscular Dystrophies|Muscular Dystrophy, Duchenne *
Approval Status: Approved
Approved Countries: Czech
Approved Indications: None
Known Adverse Events: None
Company: Santhera
Company Location:
Company Founding Year: None
Additional Commercial Interests: None
Clinical Description
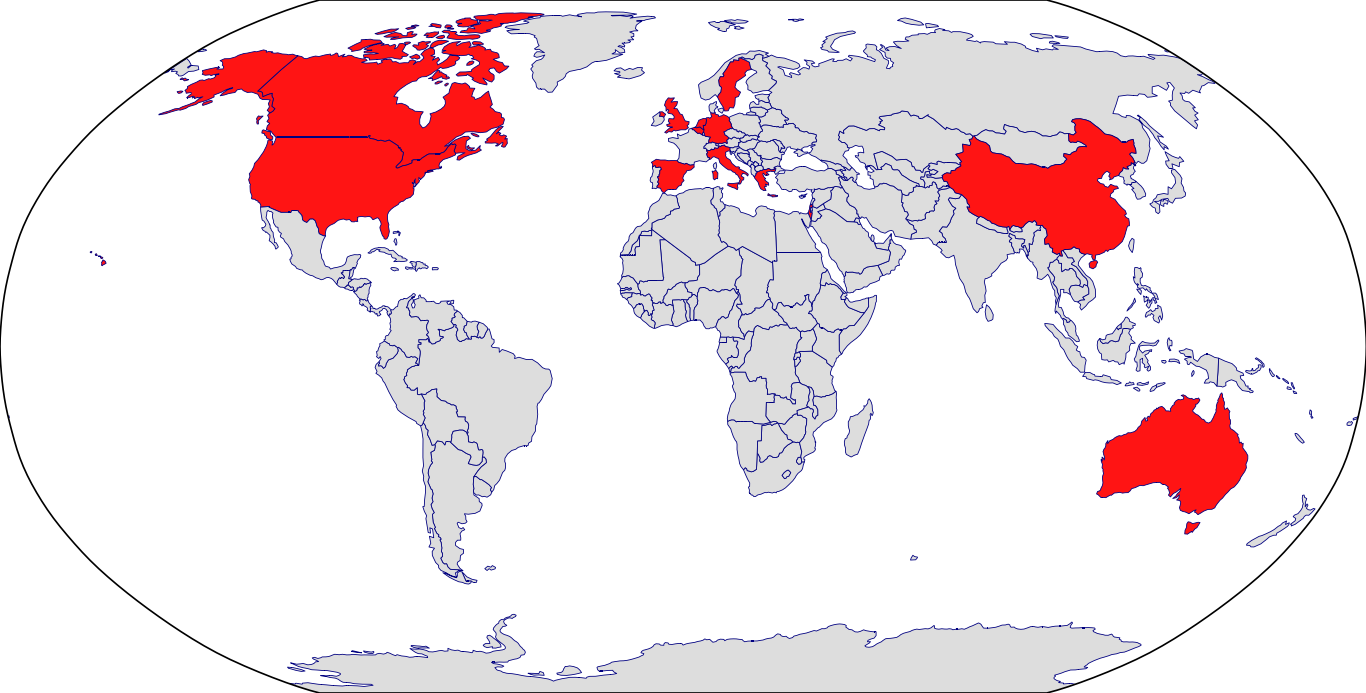
Countries in Clinic: Canada, China, Germany, Italy, United States
Active Clinical Trial Count: 7
Recent & Upcoming Milestones
- Clinical Outcomes Reported - Catalyst Biosciences presented P2 Muscular Dystrophy, Duchenne results on 2024-03-03 for Vamorolone
- Clinical Outcomes Reported - Catalyst Biosciences presented P2 Muscular Dystrophy, Duchenne results on 2024-02-21 for Vamorolone
- Catalyst Pharmaceuticals announced a PDUFA action date of October 26, 2023, for vamorolone, a promising treatment for Duchenne muscular dystrophy.
Highest Development Phases
Phase 2: Muscular Dystrophy, Becker|Muscular Dystrophy, Duchenne
Phase 1: Healthy Volunteers
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05166109 |
VBP15-BMD-001 | P2 |
Completed |
Muscular Dystrophy, Duchenne|Muscular Dystrophy, Becker |
2025-08-18 |
50% |
2025-09-19 |
|
NCT06649409 |
SNT-I-VAM-026 | P1 |
Completed |
Healthy Volunteers |
2024-06-29 |
2024-10-19 |
Primary Endpoints|Treatments |
|
CTR20232865 |
CTR20232865 | P1 |
Completed |
Muscular Dystrophy, Duchenne |
2024-01-08 |
2025-04-29 |
||
NCT06689527 |
SNT-I-VAM-025 | P1 |
Completed |
Healthy Volunteers |
2024-09-24 |
2025-12-16 |
Primary Endpoints|Treatments |
|
2022-000844-31 |
Trial of Vamorolone vs. Placebo for the Treatment of Becker Muscular Dystrophy | P2 |
Completed |
Muscular Dystrophy, Becker |
2025-05-18 |
2025-05-06 |
||
NCT05185622 |
VBP15-006 | P2 |
Completed |
Muscular Dystrophy, Duchenne |
2024-07-16 |
50% |
2025-10-28 |
|
2025-000201-16 |
006 | P2 |
Completed |
Muscular Dystrophy, Duchenne |
2024-05-07 |
2025-07-09 |
Primary Completion Date|Start Date|Study Completion Date|Treatments |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
02/10/2026 |
News Article |
Santhera Appoints Stifel as Corporate Finance Advisor |
|
01/30/2026 |
News Article |
DEFEAT DUCHENNE CANADA MARKS 30 YEARS OF RESEARCH INVESTMENT, SURPASSES $20 MILLION MILESTONE |
|
01/15/2026 |
News Article |
Santhera Receives Swissmedic Approval of AGAMREE® (Vamorolone) for the Treatment of Duchenne Muscular Dystrophy |
|
01/08/2026 |
News Article |
Santhera Licenses AGAMREE® (Vamorolone) to Nxera Pharma in Japan, South Korea, Australia and New Zealand in a Deal Valued at up to USD 205 Million Plus Royalties |


