Product Description
Mechanisms of Action: GDF8 Inhibitor
Novel Mechanism: Yes
Modality: Antibody
Route of Administration: Intravenous, Subcutaneous
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Regeneron
Company Location: Eastern America
Company Founding Year: 1988
Additional Commercial Interests: None
Clinical Description
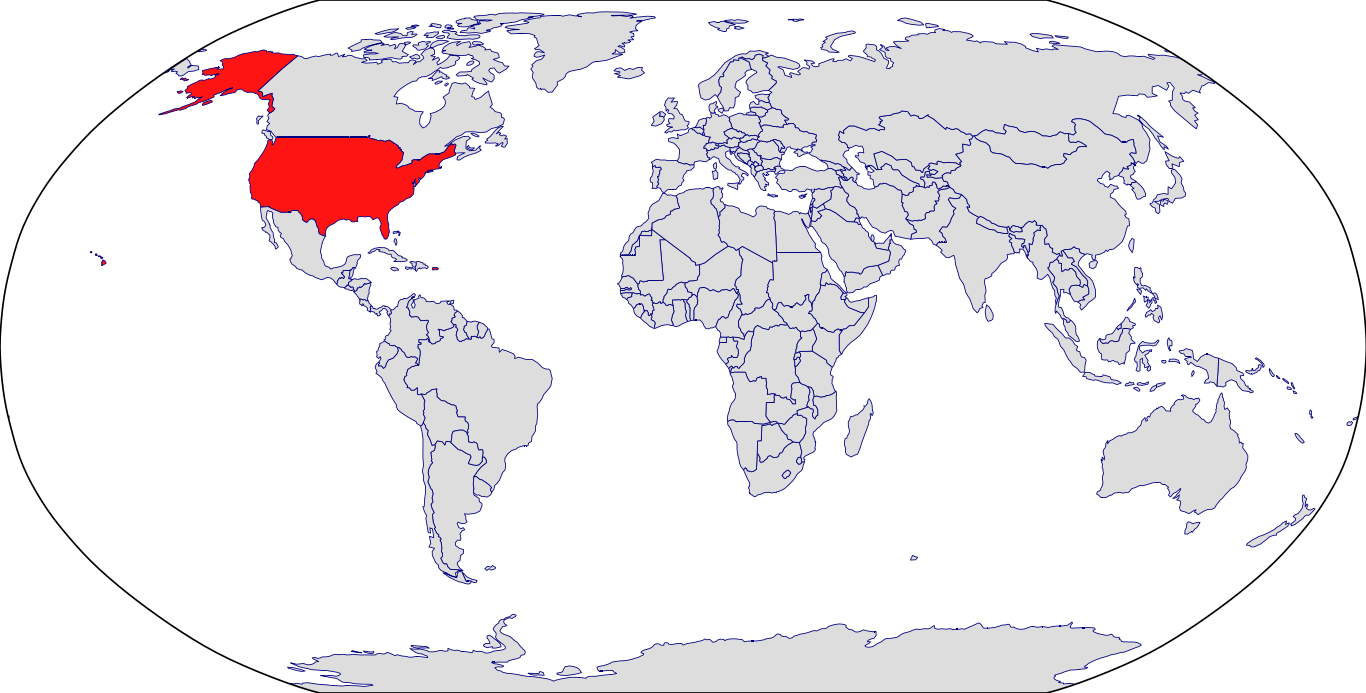
Countries in Clinic: Puerto Rico, United States
Active Clinical Trial Count: 1
Recent & Upcoming Milestones
- Clinical Outcomes Reported - Regeneron presented P2 Obesity results on 2025-06-02 for Trevogrumab
- Clinical Outcomes Reported - Regeneron announced they will present P2 Obesity results in 2H25 for Trevogrumab
Highest Development Phases
Phase 2: Obesity
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT06299098 |
COURAGE | P2 |
Active, not recruiting |
Obesity |
2026-05-18 |
50% |
2025-12-18 |
Primary Completion Date|Primary Endpoints|Study Completion Date |


