Product Description
Tranexamic acid is used to treat heavy menstrual bleeding in women. This medicine may be used by teenage females, but is not intended for use before the start of menstruation. Tranexamic acid is an antifibrinolytic agent. It works by blocking the breakdown of blood clots, which prevents bleeding. (Sourced from: https://www.mayoclinic.org/drugs-supplements/tranexamic-acid-oral-route/description/drg-20073517)
Mechanisms of Action: Fibrinolysis Inhibitor
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Intravenous, Topical, Oral
FDA Designation: *
Approval Status: Approved
Approved Countries: Argentina | Australia | Austria | Bangladesh | Belgium | Bosnia | Brazil | Bulgaria | Canada | Chile | China | Colombia | Croatia | Cyprus | Czech | Denmark | Dominican Republic | Ecuador | Egypt | Estonia | Finland | France | Germany | Greece | Hong Kong | Hungary | Iceland | India | Indonesia | Ireland | Israel | Italy | Jordan | Korea | Latvia | Lebanon | Lithuania | Luxembourg | Malaysia | Malta | Mexico | Morocco | Netherlands | New Zealand | Norway | Pakistan | Peru | Philippines | Poland | Portugal | Romania | Russia | Saudi Arabia | Serbia | Singapore | Slovakia | Slovenia | South Africa | Spain | Sri Lanka | Sweden | Switzerland | Taiwan | Thailand | Turkey | Ukraine | United Arab Emirates | United Kingdom | United States | Uruguay | Venezuela | Vietnam
Approved Indications: None
Known Adverse Events: None
Company: Ferring
Company Location: Europe
Company Founding Year: 1950
Additional Commercial Interests: None
Clinical Description
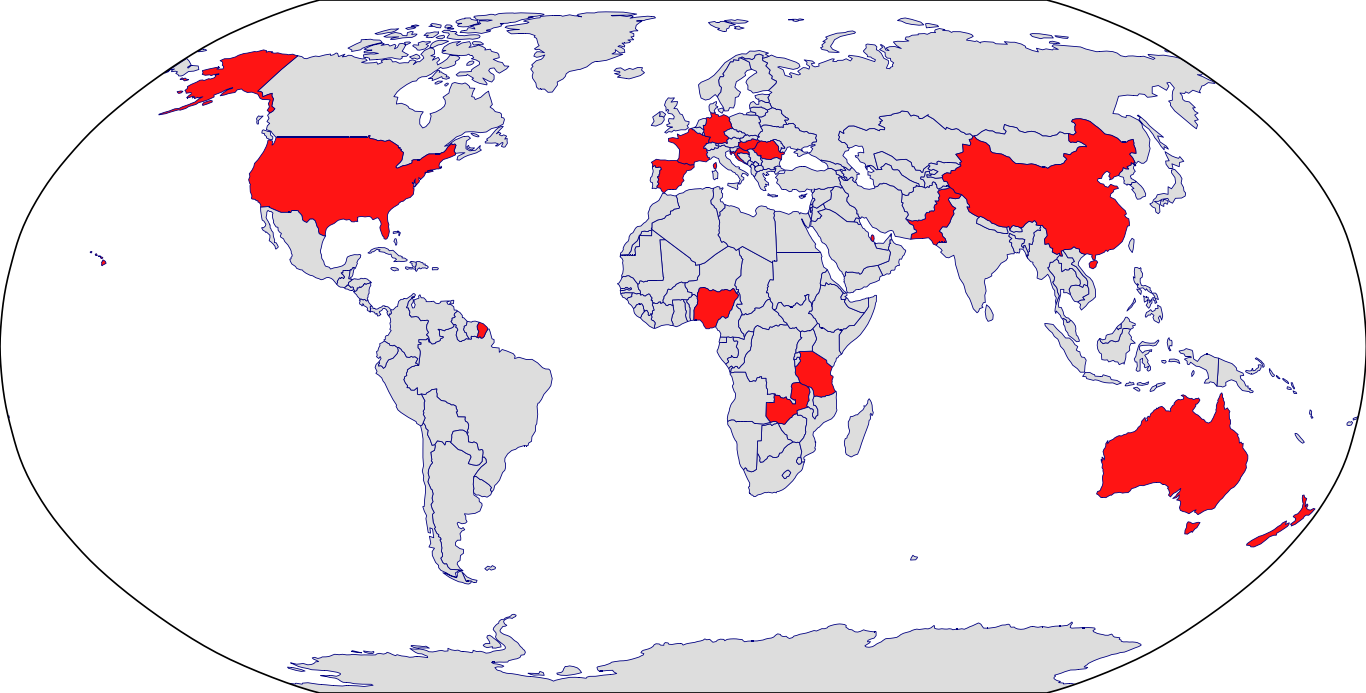
Countries in Clinic: Croatia, Germany, Hungary, Nigeria, Pakistan, Romania, Spain, Tanzania, United States, Zambia
Active Clinical Trial Count: 9
Recent & Upcoming Milestones
Highest Development Phases
Phase 3: Anemia|Brain Hemorrhage, Traumatic|Hip Fracture|Injuries/wounds Unspecified|Sarcoma
Phase 2: Stroke
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
2020-004746-10 |
STOP-MSU | P2 |
Completed |
Stroke |
2023-05-28 |
2025-05-16 |
Treatments |
|
2023-503719-13-00 |
HYL-P004-003 | P3 |
Recruiting |
Injuries/wounds Unspecified |
2024-06-27 |
4% |
2025-05-02 |
Treatments |
NCT05047133 |
NCT05047133 | P3 |
Recruiting |
Hip Fracture |
2022-07-01 |
2024-03-06 |
||
2016-003214-27 |
UTOTXA_2016 | P3 |
Active, not recruiting |
Sarcoma |
2019-01-31 |
2022-03-13 |
Treatments |
|
NCT04387305 |
TIC-TOC | P3 |
Not yet recruiting |
Brain Hemorrhage, Traumatic |
2031-03-31 |
2025-12-16 |
Primary Completion Date|Primary Endpoints|Start Date|Study Completion Date |
|
NCT06414031 |
TATRA | P3 |
Not yet recruiting |
Anemia |
2025-11-30 |
2024-05-15 |
Primary Endpoints|Treatments |
|
2005-005103-41 |
2005-005103-41 | P2 |
Completed |
Unknown |
2025-01-30 |
2025-05-06 |
Primary Completion Date|Study Completion Date|Treatments|Trial Status |
|
2018-004552-37 |
PRADO | P2 |
Active, not recruiting |
Unknown |
2022-05-07 |
2025-05-06 |
Treatments |
|
NCT03475342 |
WOMAN-2 | P3 |
Completed |
Anemia |
2023-09-20 |
2024-01-11 |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
12/09/2025 |
News Article |
Drug Reduced Need for Blood Transfusions During Hospitalization for Non-Cardiac Surgery |
|
12/03/2025 |
News Article |
Saranghae Advanced Brightening Dark Spot Repair Serum Named "Anti-Blemish Product of the Year" By Beauty Innovation |
|
11/19/2025 |
News Article |
Rafa Laboratories Awarded up to $186 Million by BARDA to Develop a Life-Saving Intramuscular TXA Injection for Prehospital Care |
|
10/28/2025 |
News Article |
Ourself Debuts Complexion Brightening Serum Featuring Patented MI9™ Peptide and Tiered-Release Vesicle™ Technology |


