Product Description
Johnson & Johnson is developing Seltorexant as a treatment for major depressive disorder with insomnia symptoms. (Sourced from: https://www.investor.jnj.com/pipeline/development-pipeline/default.aspx)
Mechanisms of Action: OX2R Antagonist
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Johnson & Johnson
Company Location: Eastern America
Company Founding Year: 1886
Additional Commercial Interests: None
Clinical Description
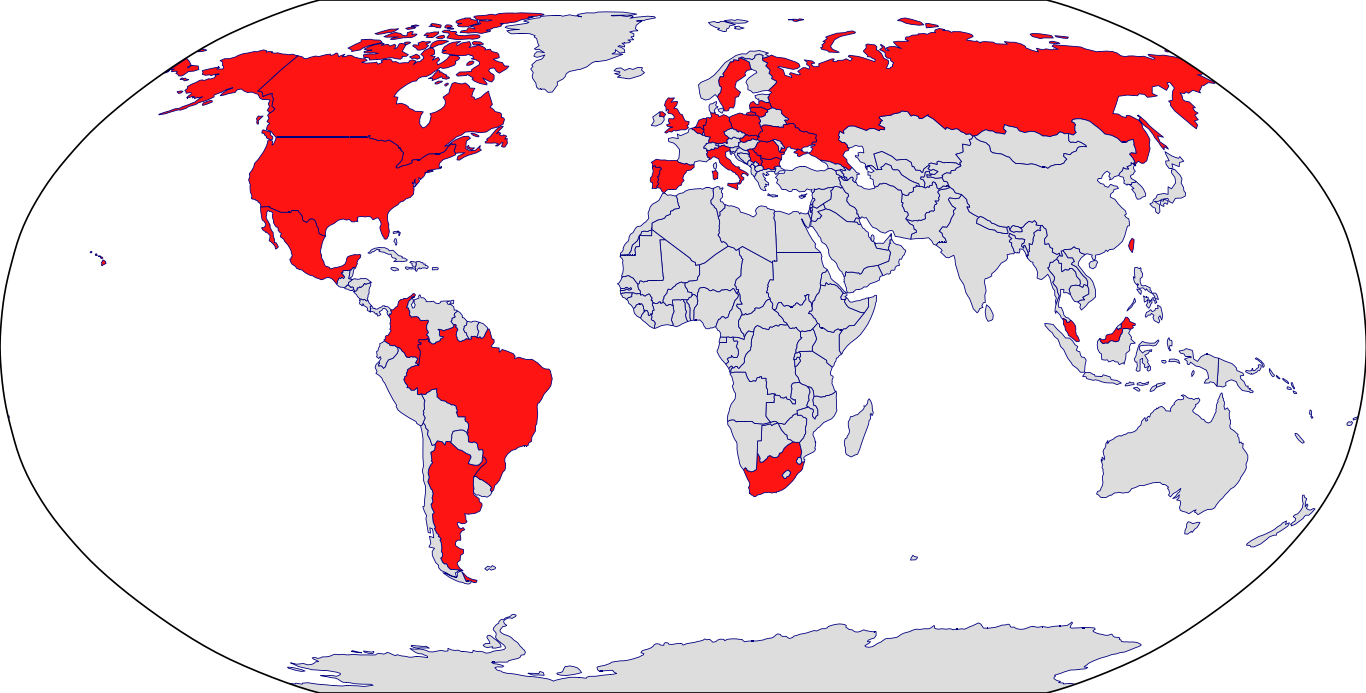
Countries in Clinic: Argentina, Belgium, Brazil, Bulgaria, Canada, Colombia, Czech Republic, Germany, Italy, Latvia, Lithuania, Malaysia, Mexico, Poland, Portugal, Romania, Russia, Serbia, Slovakia, South Africa, Spain, Sweden, Taiwan, Ukraine, United Kingdom, United States
Active Clinical Trial Count:
Recent & Upcoming Milestones
- Clinical Outcomes Reported - Johnson & Johnson presented P3 Depressive Disorder, Major results on 2026-01-14 for Seltorexant
Highest Development Phases
Phase 3: Depressive Disorder, Major|Insomnia
Phase 2: Alzheimer Disease
Phase 1: Healthy Volunteers
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
2023-509070-36-00 |
42847922MDD3003 | P3 |
Recruiting |
Insomnia|Depressive Disorder, Major |
2026-12-30 |
43% |
2025-05-02 |
Treatments |
NCT06559306 |
42847922MDD3003 | P3 |
Recruiting |
Depressive Disorder, Major |
2026-12-30 |
43% |
2024-08-20 |
|
NCT05307692 |
CR109177 | P2 |
Completed |
Alzheimer Disease |
2023-11-10 |
23% |
2024-11-27 |
Patient Enrollment|Primary Endpoints|Treatments |
2020-000337-40 |
2020-000337-40 | P3 |
Completed |
Depressive Disorder, Major |
2024-04-30 |
56% |
2025-06-30 |
Treatments |
NCT04513912 |
CR108810 | P3 |
Completed |
Depressive Disorder, Major |
2023-10-03 |
57% |
2023-12-05 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
NCT04533529 |
CR108804 | P3 |
Completed |
Depressive Disorder, Major |
2023-04-25 |
56% |
2024-05-25 |
|
2021-000567-77 |
2021-000567-77 | P1 |
Completed |
Depressive Disorder, Major |
2023-11-24 |
78% |
2025-06-24 |
Primary Completion Date|Start Date|Study Completion Date|Treatments|Trial Status |
2020-000341-14 |
2020-000341-14 | P3 |
Active, not recruiting |
Depressive Disorder, Major |
2023-01-12 |
57% |
2022-03-13 |
Treatments |
NCT05106153 |
CR109099 | P1 |
Completed |
Healthy Volunteers |
2023-05-12 |
4% |
2024-06-28 |
|
NCT04451187 |
CR108762 | P1 |
Completed |
Depressive Disorder, Major |
2023-03-16 |
64% |
2023-04-01 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |


