Product Description
Roxadustat, an oral medicine, could be the first in a new class of treatments called oral HIF-PH inhibitors that promotes erythropoiesis, or RBC production, through increased endogenous production of erythropoietin, improved iron absorption and mobilisation, and reduction of hepcidin. Roxadustat is also in clinical development for anaemia associated with MDS and for chemotherapy-induced anaemia. (Sourced from: https://www.astrazeneca.com/media-centre/press-releases/2021/update-on-us-review-of-roxadustat.html)
Mechanisms of Action: HIF Inhibitor
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: Orphan Drug - Myelodysplastic Syndrome *
Approval Status: Not Approved
Approved Countries: Austria | Bangladesh | Belgium | Chile | China | Croatia | Czech | Denmark | Egypt | Estonia | European Medicines Agency | Finland | France | Germany | Greece | Hungary | Iceland | Ireland | Italy | Japan | Korea | Latvia | Lithuania | Mexico | Netherlands | Norway | Philippines | Poland | Portugal | Romania | Russia | Saudi Arabia | Slovakia | Slovenia | South Africa | Spain | Sweden | Turkey | Ukraine | United Arab Emirates | United Kingdom
Approved Indications: None
Known Adverse Events: None
Company: FibroGen
Company Location: Western America
Company Founding Year: 1993
Additional Commercial Interests: AstraZeneca
Clinical Description
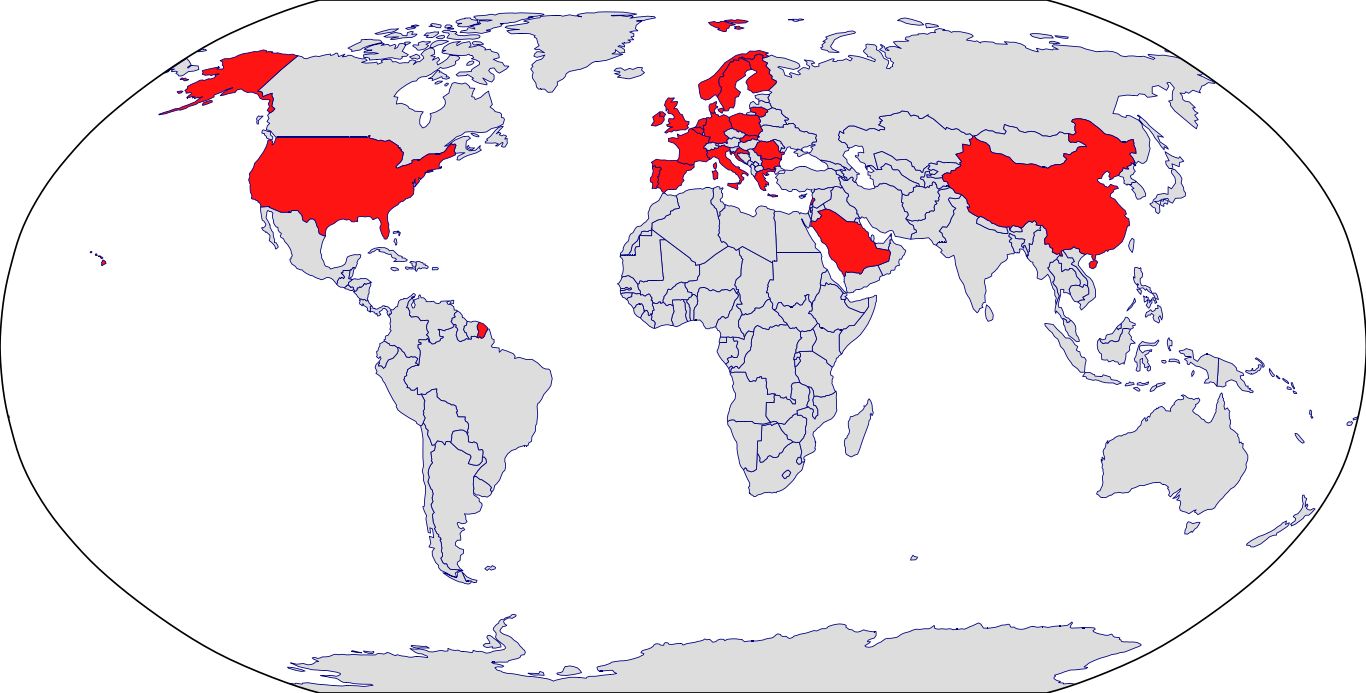
Countries in Clinic: Belgium, Bulgaria, China, Croatia, Czech Republic, Denmark, Finland, France, Germany, Greece, Ireland, Italy, Lebanon, Lithuania, Netherlands, Norway, Poland, Portugal, Romania, Saudi Arabia, Slovakia, Spain, Sweden, United Kingdom, Unknown Location
Active Clinical Trial Count: 8
Recent & Upcoming Milestones
- FDA extended review of FibroGen's NDA for anemia drug Roxadustat to March 20, 2021, aiming to address unmet patient needs.
- The pdufa date for FDA review of Roxadustat for chronic kidney disease anemia is set for December 20, 2020.
Highest Development Phases
Phase 3: Anemia|Kidney Diseases|Kidney Failure, Chronic|Myelodysplastic Syndrome|Preleukemia
Phase 1: Hyperphosphatemia
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
CTR20250363 |
CTR20250363 | P1 |
Completed |
Hyperphosphatemia|Kidney Diseases |
2025-03-28 |
2025-04-29 |
Patient Enrollment|Primary Completion Date|Start Date|Study Completion Date|Treatments|Trial Status |
|
NCT05970172 |
1517-CL-1003 | P3 |
Recruiting |
Anemia|Kidney Failure, Chronic |
2027-10-30 |
30% |
2025-11-27 |
|
2022-501980-42-00 |
1517-CL-1003 | P3 |
Recruiting |
Anemia|Kidney Diseases |
2026-06-30 |
30% |
2025-05-02 |
Treatments |
NCT03303066 |
FGCL-4592-813 | P3 |
Completed |
Myelodysplastic Syndrome|Preleukemia|Anemia |
2023-01-11 |
15% |
2023-05-04 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
NCT05301517 |
FGCL-4592-898 | P3 |
Completed |
Anemia |
2023-04-03 |
63% |
2023-06-07 |
Primary Endpoints|Study Completion Date|Treatments|Trial Status |
CTR20231278 |
CTR20231278 | P1 |
Completed |
Anemia|Kidney Diseases |
2023-07-25 |
2025-04-29 |
Patient Enrollment|Treatments |
|
2017-001773-17 |
2017-001773-17 | P3 |
Active, not recruiting |
Anemia|Myelodysplastic Syndrome |
2023-09-27 |
2022-03-13 |
Treatments |
|
CTR20213265 |
CTR20213265 | P3 |
Completed |
Anemia |
2023-04-21 |
2025-04-29 |
Patient Enrollment|Primary Completion Date|Start Date|Study Completion Date|Treatments|Trial Status |


