Product Description
Brain shuttle technology to transferAnti-CD20 antibody across the blood brain barrier and increase antibody concentrations in the brain/CNS. (Sourced from: https://www.gene.com/medical-professionals/pipeline?phase=1,2,3&category=neuroscience)
Mechanisms of Action: CD20 Inhibitor
Novel Mechanism: No
Modality: Antibody
Route of Administration: Subcutaneous, Intravenous
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Hoffmann-La Roche
Company Location:
Company Founding Year: 1896
Additional Commercial Interests: None
Clinical Description
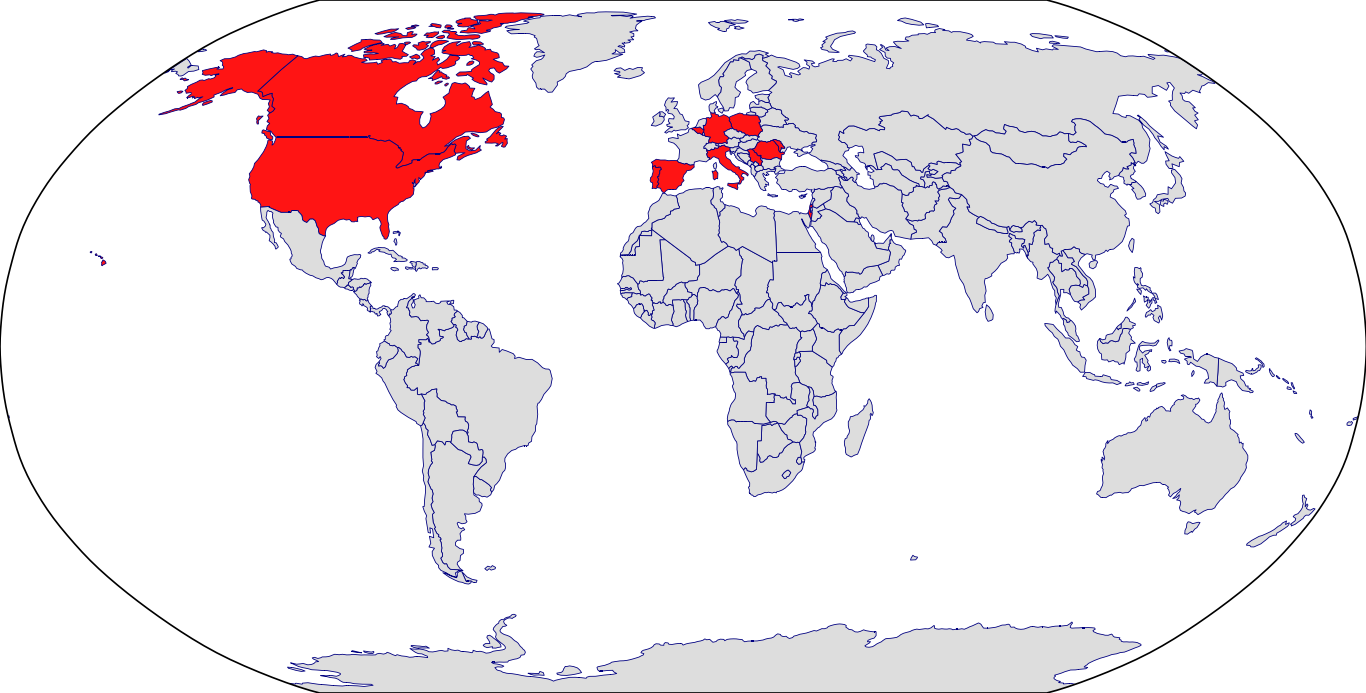
Countries in Clinic: Belgium, Canada, Germany, Israel, Italy, Moldova, Poland, Portugal, Romania, Serbia, Spain, United States
Active Clinical Trial Count: 1
Recent & Upcoming Milestones
Highest Development Phases
Phase 1: Multiple Sclerosis
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05704361 |
BP42230 | P1 |
Recruiting |
Multiple Sclerosis |
2027-07-08 |
50% |
2025-11-11 |
Recent News Events
Date |
Type |
Title |
|---|


