Product Description
Recombinant human Glutamic Acid Decarboxylase (EC 4.1.1.15, 65kDa isoform, scientific abbreviation is ÒrhGAD65Ó) expressed in baculoviral infection of Spodoptera frugiperda cells (Sourced from: https://www.diamyd.com/docs/prodfiles/rhGAD65_DIAMYD_PDS_2016-06-22.pdf)
Mechanisms of Action: Vaccine
Novel Mechanism: No
Modality: Vaccine
Route of Administration: Injection
FDA Designation: Fast Track - *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Diamyd
Company Location: Europe
Company Founding Year: None
Additional Commercial Interests: None
Clinical Description
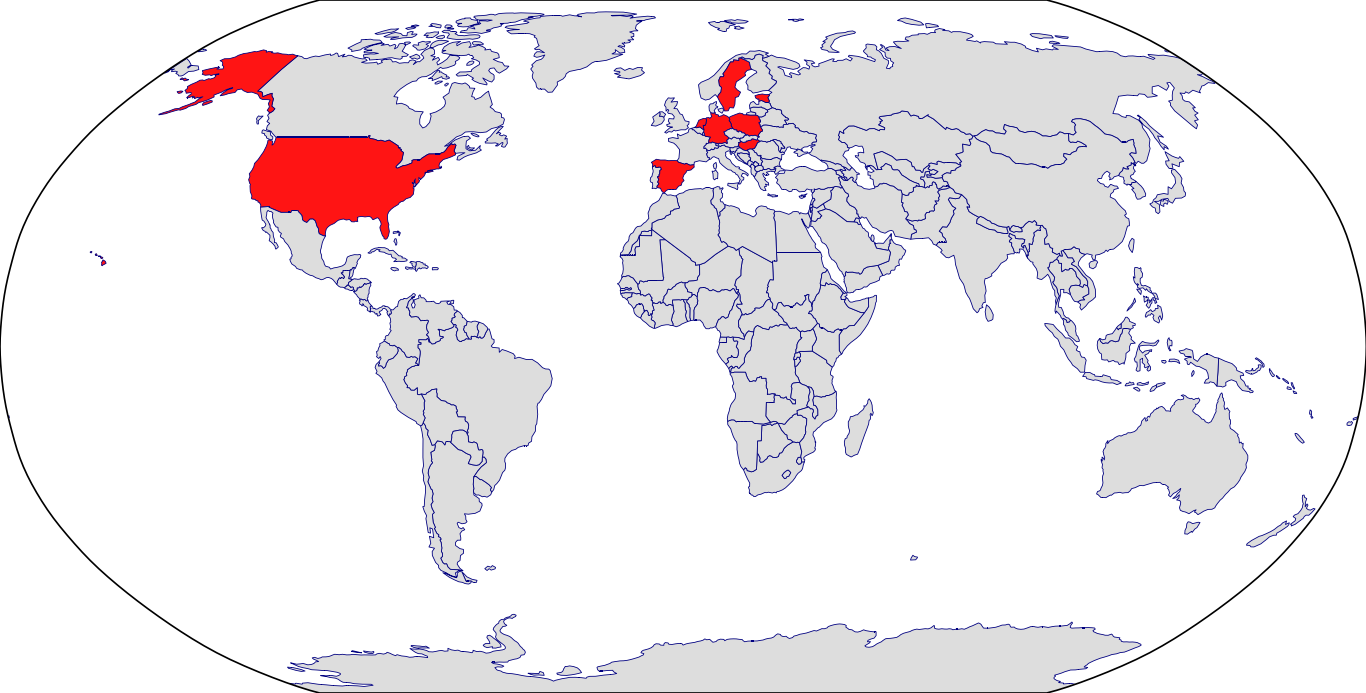
Countries in Clinic: Czech Republic, Estonia, Germany, Hungary, Netherlands, Poland, Spain, Sweden, United States
Active Clinical Trial Count: 1
Recent & Upcoming Milestones
Highest Development Phases
Phase 3: Type 1 Diabetes
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05018585 |
DIAGNODE-3 | P3 |
Active, not recruiting |
Type 1 Diabetes |
2027-12-01 |
61% |
2026-01-14 |
Primary Endpoints|Trial Status |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
02/09/2026 |
News Article |
Diamyd Medical receives Notice of Allowance for key retogatein US patent |
|
01/13/2025 |
News Article |
Diamyd Medical updates U.S. market potential for Diamyd® |
|
01/10/2025 |
News Article |
Diamyd Medical confirms key advances toward Accelerated Approval for Diamyd® following FDA Type C Meeting |
|
12/13/2024 |
News Article |
Diamyd Medical Aligns with FDA on Key Elements for an Accelerated Approval Process |


