Product Description
Mechanisms of Action: APOC3 Inhibitor
Novel Mechanism: No
Modality: Nucleic Acid
Route of Administration: N/A
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Ribocure Pharmaceuticals AB
Company Location:
Company Founding Year: None
Additional Commercial Interests: None
Clinical Description
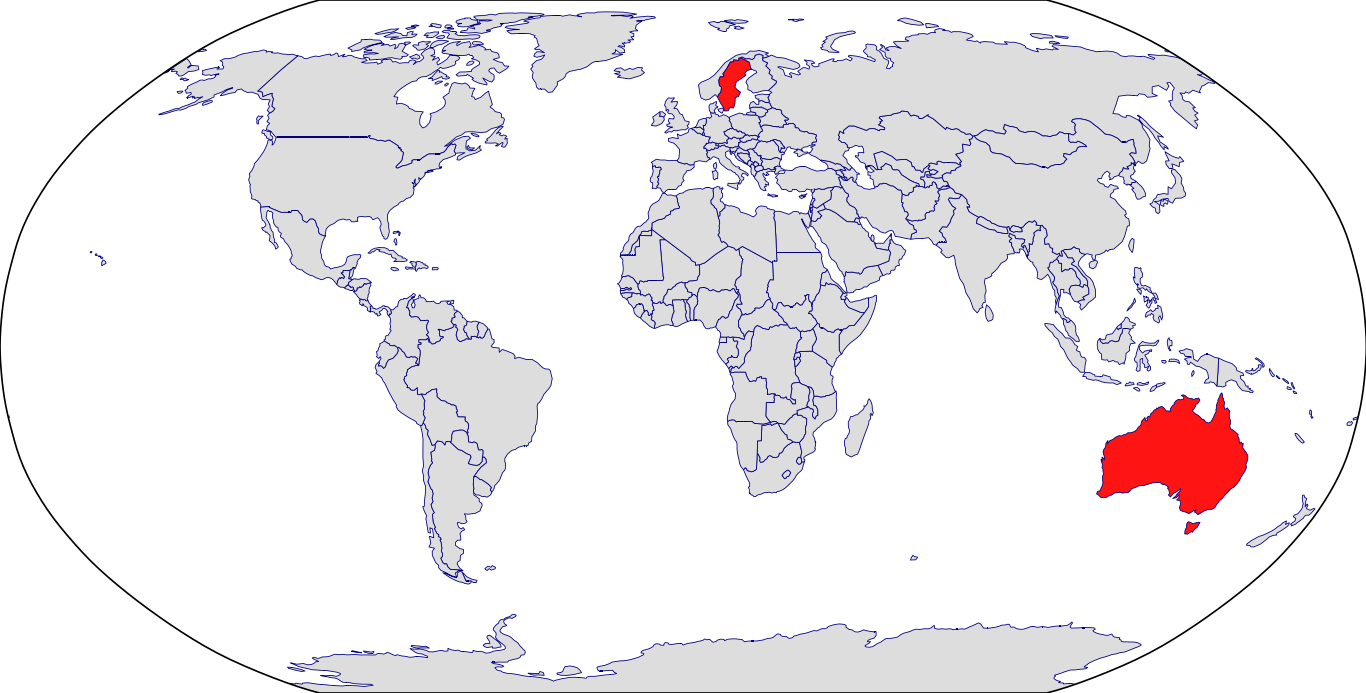
Countries in Clinic: Australia, Sweden
Active Clinical Trial Count: 3
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Dyslipidemia
Phase 1: Healthy Volunteers
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT06797401 |
RC02T001 | P2 |
Recruiting |
Dyslipidemia |
2026-05-31 |
50% |
2025-02-01 |
Primary Endpoints|Start Date|Treatments|Trial Status |
NCT05539651 |
RBAP1101 | P1 |
Completed |
Healthy Volunteers |
2024-10-30 |
69% |
2024-12-05 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
2023-510369-92-00 |
RC02T001 | P2 |
Recruiting |
Dyslipidemia |
2026-06-30 |
2025-05-02 |
Treatments |
Recent News Events
Date |
Type |
Title |
|---|


