Product Description
An orally available inhibitor of lysine specific demethylase 1 (LSD1), with potential antineoplastic activity. Upon administration, pulrodemstat binds to and inhibits LSD1, a demethylase that suppresses the expression of target genes by converting the di- and mono-methylated forms of lysine at position 4 of histone H3 (H3K4) to mono- and unmethylated H3K4, respectively. LSD1 inhibition enhances H3K4 methylation and increases the expression of tumor (remove hyphen) suppressor genes. This may lead to an inhibition of cell growth in LSD1-overexpressing tumor cells. In addition, LSD1 demethylates mono- or di-methylated H3K9 which increases gene expression of tumor promoting genes; inhibition of LSD1 promotes H3K9 methylation and decreases transcription of these genes. LSD1, an enzyme belonging to the flavin adenine dinucleotide (FAD)-dependent amine oxidase family that is overexpressed in certain tumor cells, plays a key role in tumor cell growth and survival. (Sourced from: https://www.cancer.gov/publications/dictionaries/cancer-drug/def/pulrodemstat)
Mechanisms of Action: LSD1 Inhibitor
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Bristol-Myers Squibb
Company Location: Eastern America
Company Founding Year: 1989
Additional Commercial Interests: None
Clinical Description
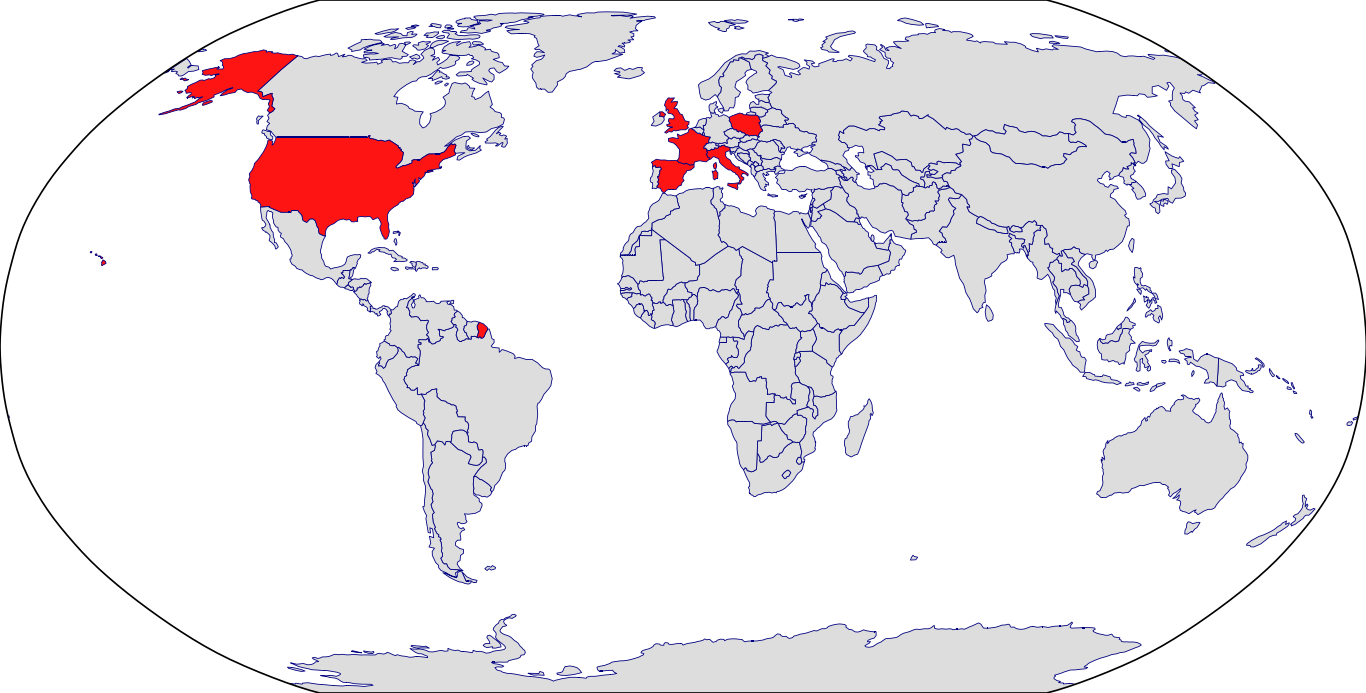
Countries in Clinic: France, Italy, Poland, Spain, United Kingdom, United States
Active Clinical Trial Count: 5
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Non-Small-Cell Lung Cancer|Small Cell Lung Cancer
Phase 1: Lymphoma, Non-Hodgkin|Prostate Cancer
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
jRCT2031210081 |
jRCT2031210081 | P1 |
Active, not recruiting |
Lymphoma, Non-Hodgkin |
2024-11-30 |
|||
NCT03850067 |
CC-90011-SCLC-001 | P1 |
Completed |
Small Cell Lung Cancer |
2024-07-16 |
21% |
2024-11-27 |
Primary Endpoints|Treatments |
NCT04628988 |
CC-90011-PCA-001 | P1 |
Completed |
Prostate Cancer |
2023-05-26 |
12% |
2023-09-14 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
NCT04350463 |
CC-90011-ST-002 | P2 |
Completed |
Non-Small-Cell Lung Cancer|Small Cell Lung Cancer |
2023-12-19 |
12% |
2024-02-22 |
Primary Endpoints|Treatments|Trial Status |
2019-004194-95 |
2019-004194-95 | P2 |
Completed |
Non-Small-Cell Lung Cancer |
2024-02-19 |
12% |
2025-05-06 |
Primary Completion Date|Study Completion Date|Treatments |


