Product Description
Mechanisms of Action: Imaging Agent
Novel Mechanism: No
Modality: Diagnostic Agent
Route of Administration: N/A
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Ratio Therapeutics, Inc.
Company Location: Eastern America
Company Founding Year: None
Additional Commercial Interests: None
Clinical Description
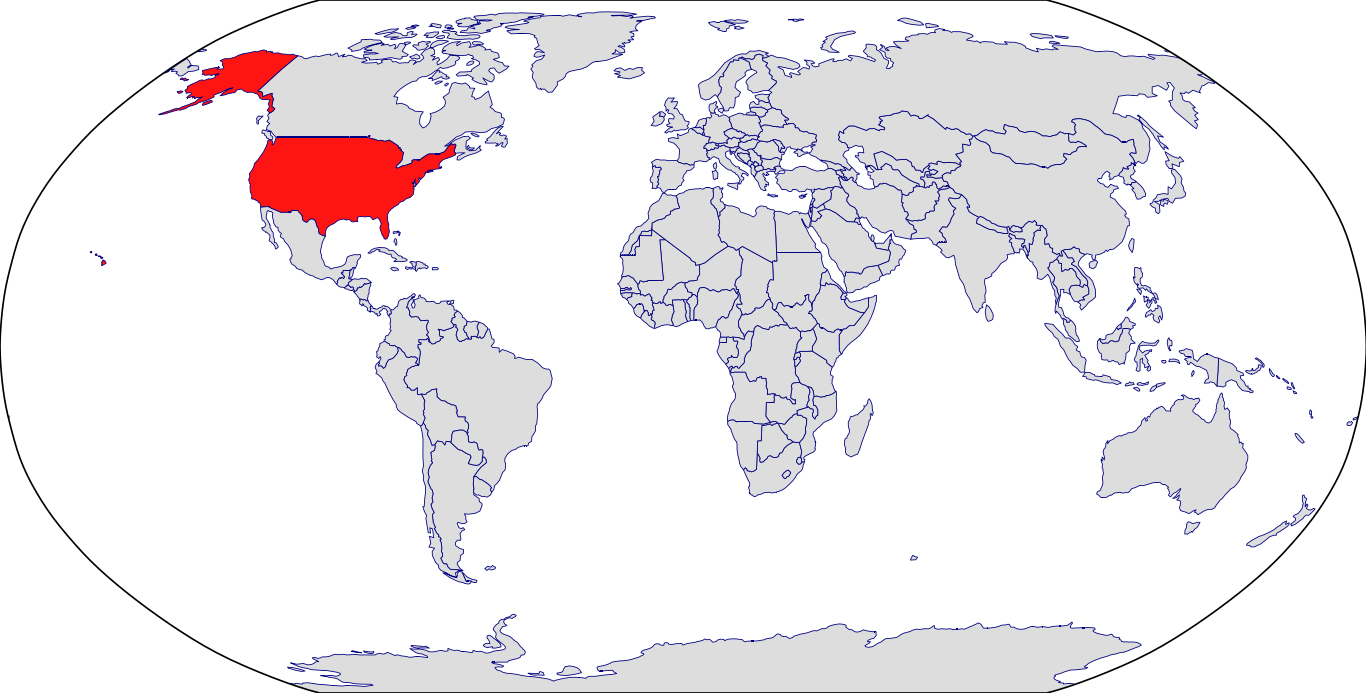
Countries in Clinic: United States
Active Clinical Trial Count: 1
Recent & Upcoming Milestones
Highest Development Phases
Phase 1: Prostate Cancer
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05773703 |
RTX-PSMA-P101 | P1 |
Completed |
Prostate Cancer |
2023-12-12 |
2023-12-14 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
Recent News Events
Date |
Type |
Title |
|---|


