Product Description
PF-06835375 is a selective, humanized, afucosyl Ig G1 antibody against CXCR5. (Sourced from: https://www.clinicaltrialsregister.eu/ctr-search/trial/2021-002897-19/HU)
Mechanisms of Action: CXCR5 Antagonist
Novel Mechanism: Yes
Modality: Antibody
Route of Administration: Intravenous, Subcutaneous
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Pfizer
Company Location: Eastern America
Company Founding Year: 1849
Additional Commercial Interests: None
Clinical Description
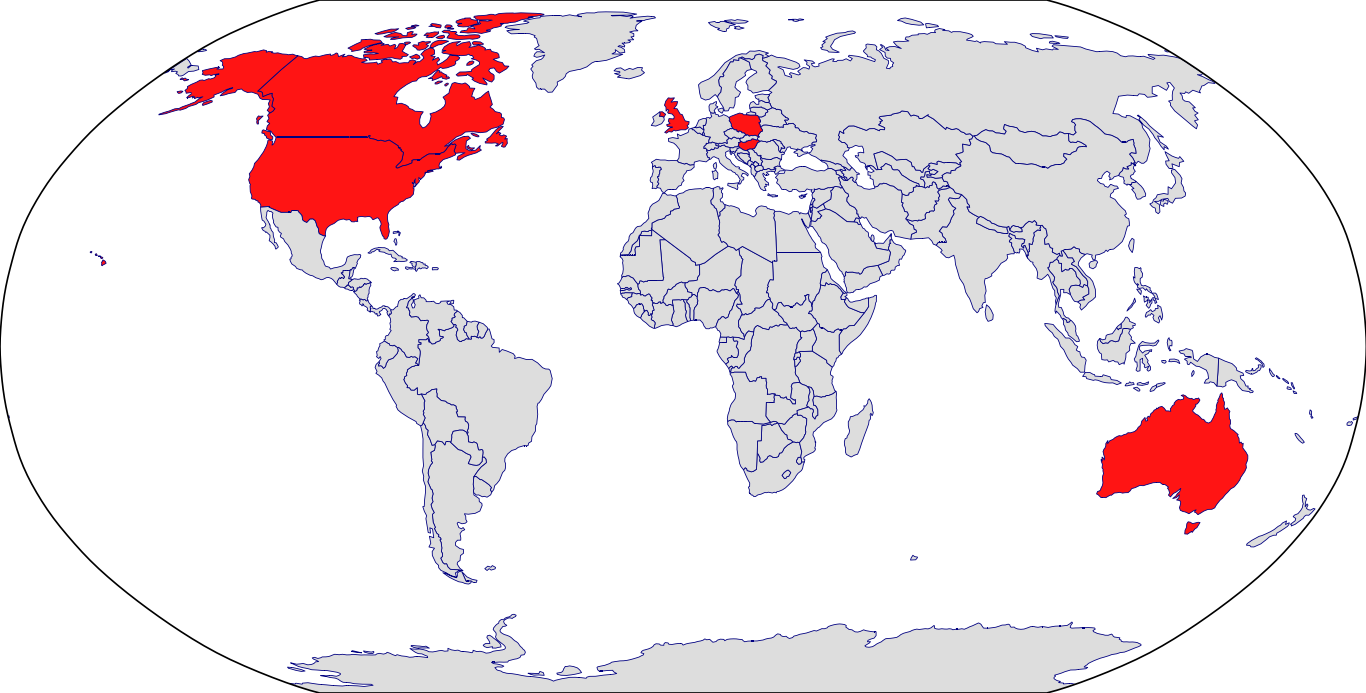
Countries in Clinic: Australia, Canada, Czech Republic, Hungary, Poland, United Kingdom, United States
Active Clinical Trial Count: 3
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Purpura, Thrombocytopenic, Idiopathic|Thrombocytopenia
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05070845 |
C1131003 | P2 |
Recruiting |
Thrombocytopenia|Purpura, Thrombocytopenic, Idiopathic |
2027-10-01 |
50% |
2025-05-22 |
Primary Completion Date|Primary Endpoints|Study Completion Date |
2023-509338-21-00 |
C1131003 | P2 |
Temporary halt |
Thrombocytopenia|Purpura, Thrombocytopenic, Idiopathic |
2026-10-27 |
50% |
2025-05-02 |
Treatments |
2021-002897-19 |
A Phase 2 Open-Label Safety and Efficacy Study of PF-06835375 | P2 |
Active, not recruiting |
Purpura, Thrombocytopenic, Idiopathic|Thrombocytopenia |
2024-12-24 |
2022-03-13 |
Treatments |
Recent News Events
Date |
Type |
Title |
|---|


