Product Description
Patisiran, an investigational RNA interference therapeutic agent, specifically inhibits hepatic synthesis of transthyretin. (Sourced from: https://pubmed.ncbi.nlm.nih.gov/29972753/)
Mechanisms of Action: TTR Modulator
Novel Mechanism: No
Modality: Nucleic Acid
Route of Administration: Intravenous
FDA Designation: *
Approval Status: Approved
Approved Countries: Australia | Austria | Belgium | Brazil | Canada | Croatia | Cyprus | Czech | Denmark | Estonia | European Medicines Agency | Finland | France | Germany | Greece | Hungary | Iceland | Ireland | Israel | Italy | Japan | Latvia | Lithuania | Luxembourg | Netherlands | Poland | Portugal | Romania | Slovakia | Slovenia | Spain | Sweden | Switzerland | Taiwan | United Kingdom | United States
Approved Indications: None
Known Adverse Events: None
Company: Alnylam
Company Location: Eastern America
Company Founding Year: 2002
Additional Commercial Interests: None
Clinical Description
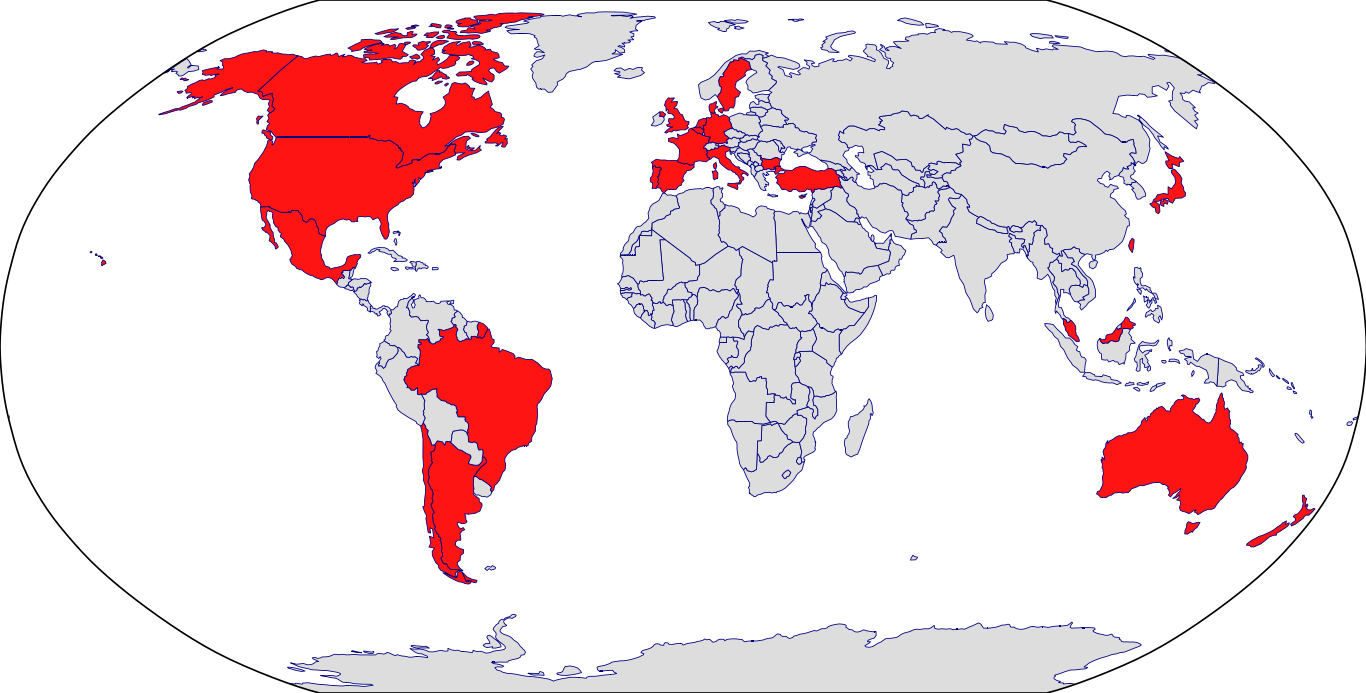
Countries in Clinic: Argentina, Australia, Belgium, Brazil, Bulgaria, Chile, Czech Republic, Denmark, France, Hong Kong, Italy, Japan, Korea, Mexico, Netherlands, New Zealand, Portugal, Sweden, Taiwan, United Kingdom, United States, Unknown Location
Active Clinical Trial Count: 5
Recent & Upcoming Milestones
- Clinical Outcomes Reported - Alnylam presented P3 Amyloidosis results on 2024-08-30 for Patisiran
- PDUFA date: FDA set October 8, 2023 for reviewing patisiran treatment for cardiomyopathy of transthyretin-mediated amyloidosis.
Highest Development Phases
Phase 3: Amyloid Neuropathies, Familial|Amyloidosis|Amyloidosis, Familial|Cardiomyopathies|Transthyretin Amyloidosis
Phase 1: Polyneuropathies
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05023889 |
001 amendment 02 | P1 |
Active, not recruiting |
Transthyretin Amyloidosis|Amyloid Neuropathies, Familial|Amyloidosis, Familial|Polyneuropathies |
2026-02-19 |
50% |
2025-10-04 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments |
jRCT2031210085 |
jRCT2031210085 | P3 |
Active, not recruiting |
Amyloidosis |
2025-07-31 |
|||
NCT03997383 |
APOLLO-B | P3 |
Active, not recruiting |
Amyloid Neuropathies, Familial|Transthyretin Amyloidosis|Cardiomyopathies|Amyloidosis, Familial |
2022-06-20 |
26% |
2024-05-25 |
|
2023-508364-29-00 |
ALN-TTR02-011 | P3 |
Recruiting |
Transthyretin Amyloidosis |
2025-06-30 |
2025-05-02 |
Treatments |
|
JapicCTI-184235 |
JapicCTI-184235 | P3 |
Active |
Transthyretin Amyloidosis |
2022-08-01 |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
03/03/2026 |
News Article |
Protego Biopharma Appoints John M. Maraganore, Ph.D., to Board of Directors |
|
03/03/2026 |
News Article |
Roivant Announces Genevant Sciences' and Arbutus Biopharma's $2.25 Billion Global Settlement With Moderna |
|
02/12/2026 |
News Article |
Alnylam Pharmaceuticals Reports Fourth Quarter and Full Year 2025 Financial Results and Highlights Recent Period Progress |
|
01/08/2026 |
News Article |
Acuitas Therapeutics Expands Collaboration with RNA Technologies & Therapeutics Through Strategic Investment |


