Product Description
VONJO is indicated for the treatment of adults with intermediate or high-risk primary or secondary (post-polycythemia vera or post-essential thrombocythemia) myelofibrosis (MF) with a platelet count below 50 × 109/L. (Sourced from: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=4b5ab444-0e1a-4984-99db-76ad11a298ee)
Mechanisms of Action: JAK2 Inhibitor, IRAK1 Inhibitor
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: Accelerated Approval - Myelofibrosis|Thrombocytopenia *
Approval Status: Approved
Approved Countries: United States
Approved Indications: None
Known Adverse Events: None
Company: CTI BioPharma
Company Location:
Company Founding Year: 1991
Additional Commercial Interests: None
Clinical Description
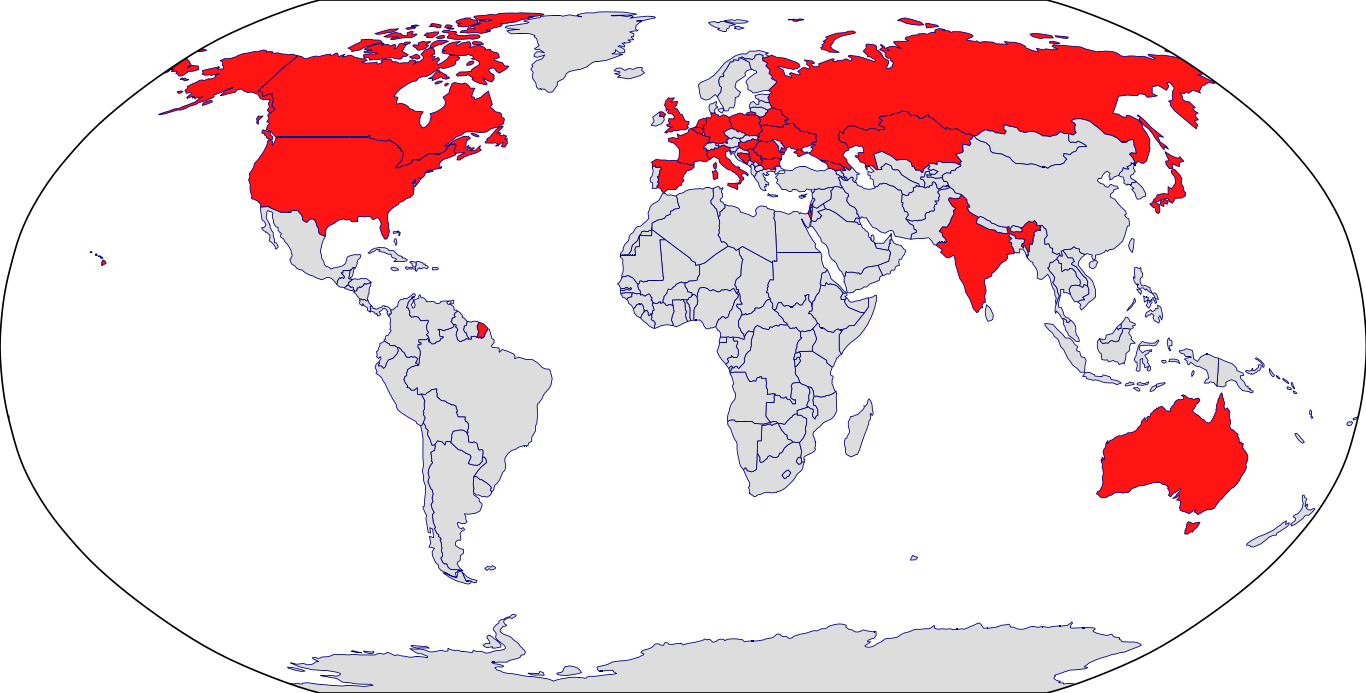
Countries in Clinic: Australia, Belarus, Belgium, Bosnia, Bulgaria, Canada, Czech Republic, France, Georgia, Germany, Hungary, India, Israel, Italy, Japan, Kazakhstan, Korea, Netherlands, Poland, Romania, Russia, Serbia, Spain, Ukraine, United Kingdom, United States, Unknown Location
Active Clinical Trial Count: 15
Recent & Upcoming Milestones
- Summary: CTI Biopharma Corp. is working towards a new therapeutic option for cytopenic myelofibrosis patients by the February 28, 2021, PDUFA data.
- The PDUFA date for pacritinib's new drug application was extended to February 28, 2022, due to additional clinical data submission.
- CTI Biopharma awaits FDA decision on Pacritinib for myelofibrosis, with a PDUFA target action date of November 30, 2021.
Highest Development Phases
Phase 3: Myelofibrosis|Polycythemia Vera|Thrombocythemia, Essential|Thrombocytosis
Phase 2: Allogeneic Stem Cell Transplant|Anemia|Bone Cancer|Bone Marrow Diseases|Bone Marrow Transplantation|Chronic Myelomonocytic Leukemia|Hematopoietic Stem Cell Transplant|Myelodysplastic Syndrome|Myeloproliferative Disorders|Thrombocytopenia
Phase 1: Healthy Volunteers|Hepatic Insufficiency
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT06538181 |
NCT06538181 | P1 |
Recruiting |
Thrombocytopenia|Myelofibrosis |
2027-03-31 |
50% |
2025-07-17 |
Primary Endpoints|Treatments |
NCT05552183 |
PAC110 | P1 |
Completed |
Hepatic Insufficiency |
2024-06-10 |
50% |
2024-10-22 |
Primary Endpoints |
NCT07447817 |
ILLUMINATE | P2 |
Not yet recruiting |
Polycythemia Vera|Thrombocythemia, Essential|Anemia|Thrombocytopenia|Myelofibrosis |
2030-05-24 |
50% |
2026-03-05 |
|
NCT07148947 |
RG1125470 | P2 |
Recruiting |
Bone Cancer|Bone Marrow Transplantation|Allogeneic Stem Cell Transplant|Hematopoietic Stem Cell Transplant |
2028-12-31 |
12% |
2026-02-21 |
Primary Endpoints|Start Date|Treatments|Trial Status |
NCT07033598 |
PROSPERA | P2 |
Recruiting |
Chronic Myelomonocytic Leukemia |
2028-07-01 |
12% |
2026-02-06 |
Primary Endpoints|Start Date|Treatments|Trial Status |
NCT07387354 |
MDS | P2 |
Not yet recruiting |
Myeloproliferative Disorders|Bone Marrow Diseases|Myelodysplastic Syndrome |
2027-01-01 |
50% |
2026-02-05 |
|
NCT06782373 |
PAXIS | P2 |
Active, not recruiting |
Myelodysplastic Syndrome |
2026-12-30 |
12% |
2025-12-23 |
Primary Completion Date|Primary Endpoints|Study Completion Date |
NCT03165734 |
PACIFICA | P3 |
Recruiting |
Myelofibrosis|Thrombocytosis|Thrombocythemia, Essential|Polycythemia Vera |
2026-12-31 |
33% |
2025-01-30 |
|
NCT07394153 |
PACRIMYEL | P2 |
Not yet recruiting |
Myelofibrosis |
2027-12-01 |
50% |
2026-02-10 |
Primary Endpoints|Treatments |
NCT05657613 |
PAC109 | P1 |
Completed |
Healthy Volunteers |
2023-06-08 |
69% |
2024-07-23 |
Primary Endpoints |
2024-516347-41-00 |
PAC601 | P2 |
Not yet recruiting |
Unknown |
2027-08-26 |
12% |
2025-05-02 |
Treatments |
jRCT2031250172 |
jRCT2031250172 | P2 |
Recruiting |
Unknown |
2027-06-30 |
|||
NCT03645824 |
HOVON134MF | P2 |
Active, not recruiting |
Myelofibrosis |
2023-07-01 |
36% |
2022-08-23 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
2024-515953-52-00 |
PAC303 | P3 |
Recruiting |
Myelofibrosis |
2027-06-30 |
2025-05-02 |
Treatments |
|
jRCT2031240668 |
jRCT2031240668 | P3 |
Recruiting |
Myelofibrosis |
2027-07-31 |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
11/07/2025 |
News Article |
Shinobi Therapeutics Announces the Appointment of Dr. Laurent Fischer as Chair of its Board of Directors |
|
10/20/2025 |
News Article |
DRI Healthcare Trust Announces Acquisition of a Synthetic Royalty Interest in the U.S. Sales of Veligrotug and VRDN-003 for the Treatment of Thyroid Eye Disease (TED) |
|
10/16/2025 |
News Article |
DRI Healthcare to Host Third Quarter 2025 Earnings Call and Webcast on November 6, 2025 |
|
09/19/2025 |
News Article |
DRI Healthcare Trust Promotes Zaheed Mawani to Chief Financial Officer |


