Product Description
NKT-2152 is being developed by NiKang Therapeutics for the treatment of various carinomas. (Sourced from: https://clinicaltrials.gov/ct2/show/NCT05119335?term=NKT-2152&draw=2&rank=1)
Mechanisms of Action: HIF Inhibitor
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Jiangsu Hansoh
Company Location: Asia Pacific
Company Founding Year: 1995
Additional Commercial Interests: NiKang Therapeutics
Clinical Description
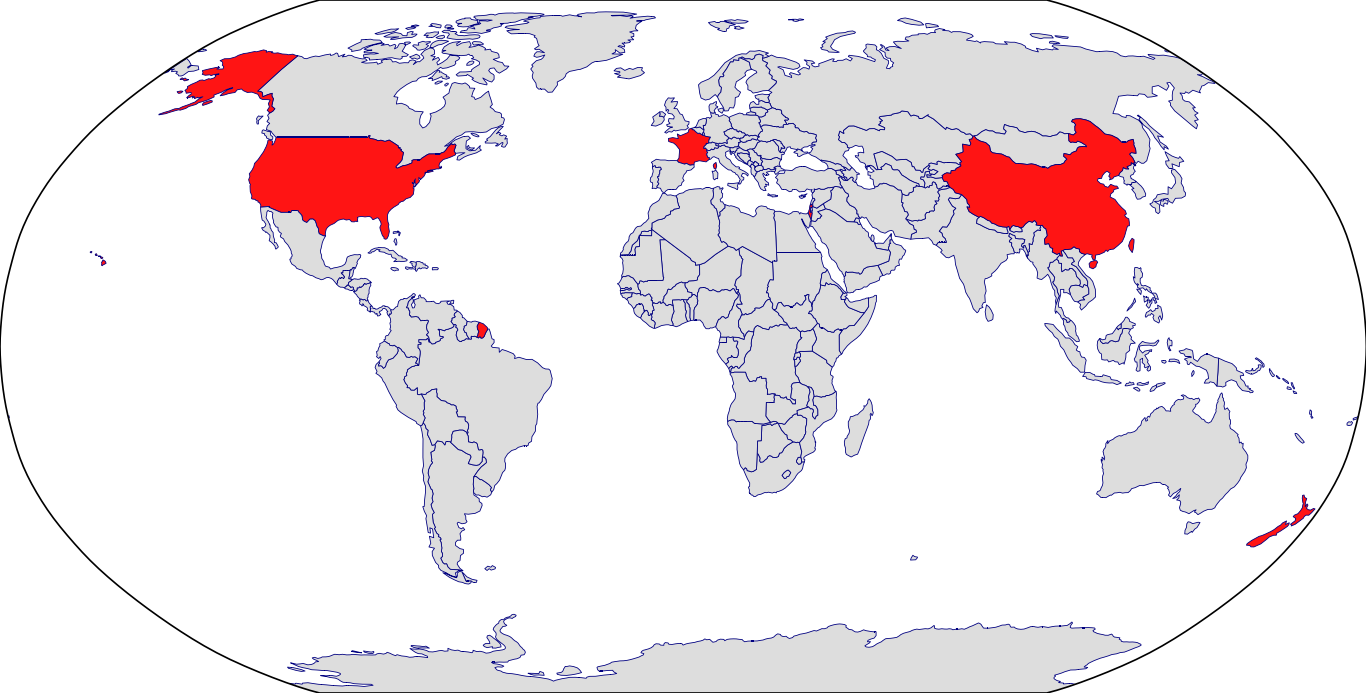
Countries in Clinic: China, France, Israel, New Zealand, South Korea, Taiwan, United States
Active Clinical Trial Count: 1
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Liver Cancer
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT04524871 |
MORPHEUS-LIVER | P2 |
Recruiting |
Liver Cancer |
2027-12-31 |
12% |
2026-02-03 |
Primary Completion Date|Primary Endpoints|Start Date|Study Completion Date |


