Product Description
Mechanisms of Action: Immunostimulant
Novel Mechanism: Yes
Modality: Peptide/Protein
Route of Administration: Inhalant
FDA Designation: *
Approval Status: Not Approved
Approved Countries: Argentina | Chile | China | Germany | Ireland | Italy | New Zealand | Pakistan | Peru | Portugal | Russia | Slovenia | Sweden | United Kingdom | Venezuela
Approved Indications: None
Known Adverse Events: None
Company: Savara
Company Location: Western America
Company Founding Year: 2007
Additional Commercial Interests: None
Clinical Description
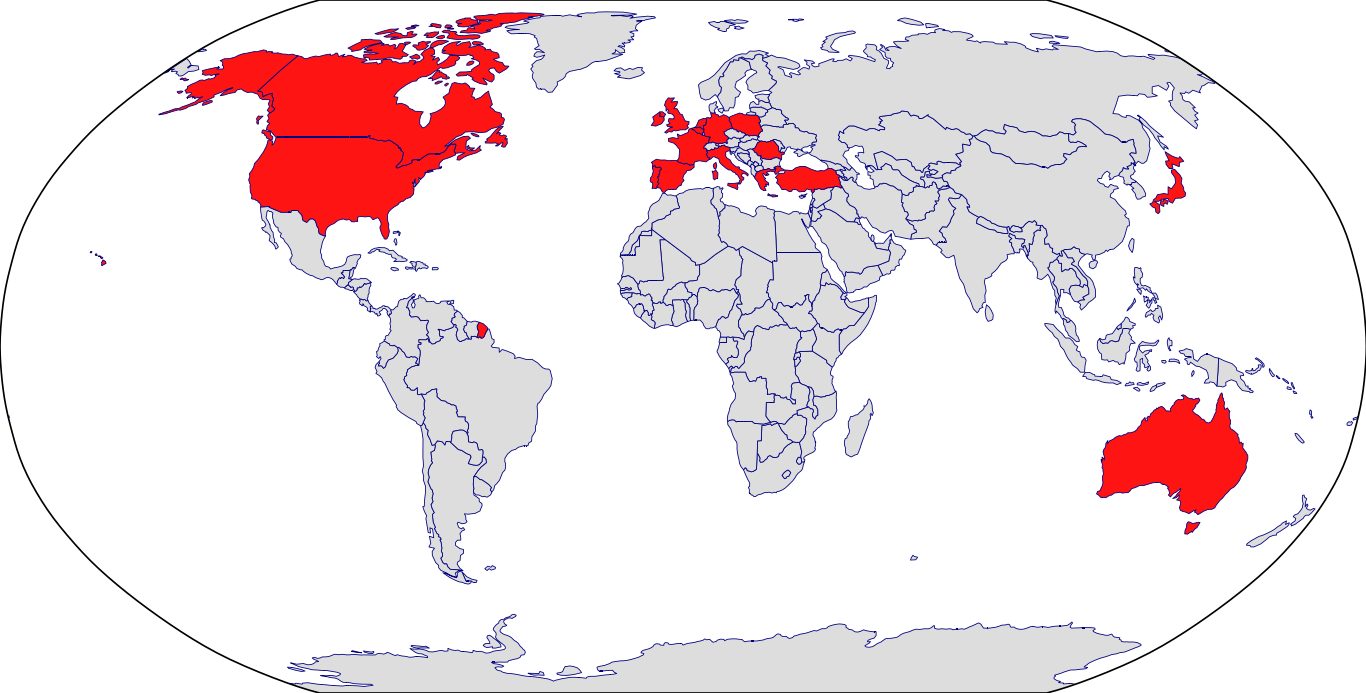
Countries in Clinic: Australia, Belgium, Canada, France, Germany, Greece, Ireland, Italy, Japan, Korea, Netherlands, Poland, Portugal, Romania, Spain, Turkey, United Kingdom, United States, Unknown Location
Active Clinical Trial Count: 6
Recent & Upcoming Milestones
- Clinical Outcomes Reported - Savara presented P3 Pulmonary Alveolar Proteinosis results on 2025-10-21 for Molgramostim
- Clinical Outcomes Reported - Savara presented P3 Pulmonary Alveolar Proteinosis results on 2025-09-29 for Molgramostim
- Clinical Outcomes Reported - Savara presented P3 Pulmonary Alveolar Proteinosis results on 2025-08-21 for Molgramostim
Highest Development Phases
Phase 3: Pulmonary Alveolar Proteinosis
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT06431776 |
aPAP | P3 |
Recruiting |
Pulmonary Alveolar Proteinosis |
2027-06-01 |
13% |
2026-02-20 |
Primary Completion Date|Primary Endpoints|Start Date|Study Completion Date |
2024-512039-66-00 |
SAV006-04 | P3 |
Not yet recruiting |
Pulmonary Alveolar Proteinosis |
2027-08-31 |
2025-04-26 |
Treatments |
|
2024-511052-41-00 |
SAV00605 | P3 |
Active, not recruiting |
Pulmonary Alveolar Proteinosis |
2026-12-31 |
2025-05-02 |
Treatments |
|
jRCT2051210036 |
jRCT2051210036 | P3 |
Recruiting |
Pulmonary Alveolar Proteinosis |
2024-12-31 |
|||
NCT04544293 |
IMPALA-2 | P3 |
Active, not recruiting |
Pulmonary Alveolar Proteinosis |
2023-11-30 |
21% |
2025-08-08 |
Primary Endpoints |
2020-001263-85 |
IMPALA-2 | P3 |
Completed |
Pulmonary Alveolar Proteinosis |
2023-10-19 |
21% |
2025-07-09 |
Treatments |


