Product Description
Maribavir is a benzimidazole riboside with activity against cytomegalovirus (CMV). (Sourced from: https://www.nejm.org/doi/full/10.1056/NEJMoa1714656)
Mechanisms of Action: pUL97 Inhibitor
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Approved
Approved Countries: Australia | Austria | Belgium | Croatia | Czech | Denmark | Estonia | European Medicines Agency | Finland | France | Germany | Greece | Hungary | Iceland | Ireland | Israel | Italy | Korea | Latvia | Lithuania | Norway | Poland | Portugal | Romania | Slovakia | Slovenia | Spain | Sweden | United Kingdom | United States
Approved Indications: None
Known Adverse Events: None
Company: Takeda
Company Location: Asia Pacific
Company Founding Year: 1781
Additional Commercial Interests: None
Clinical Description
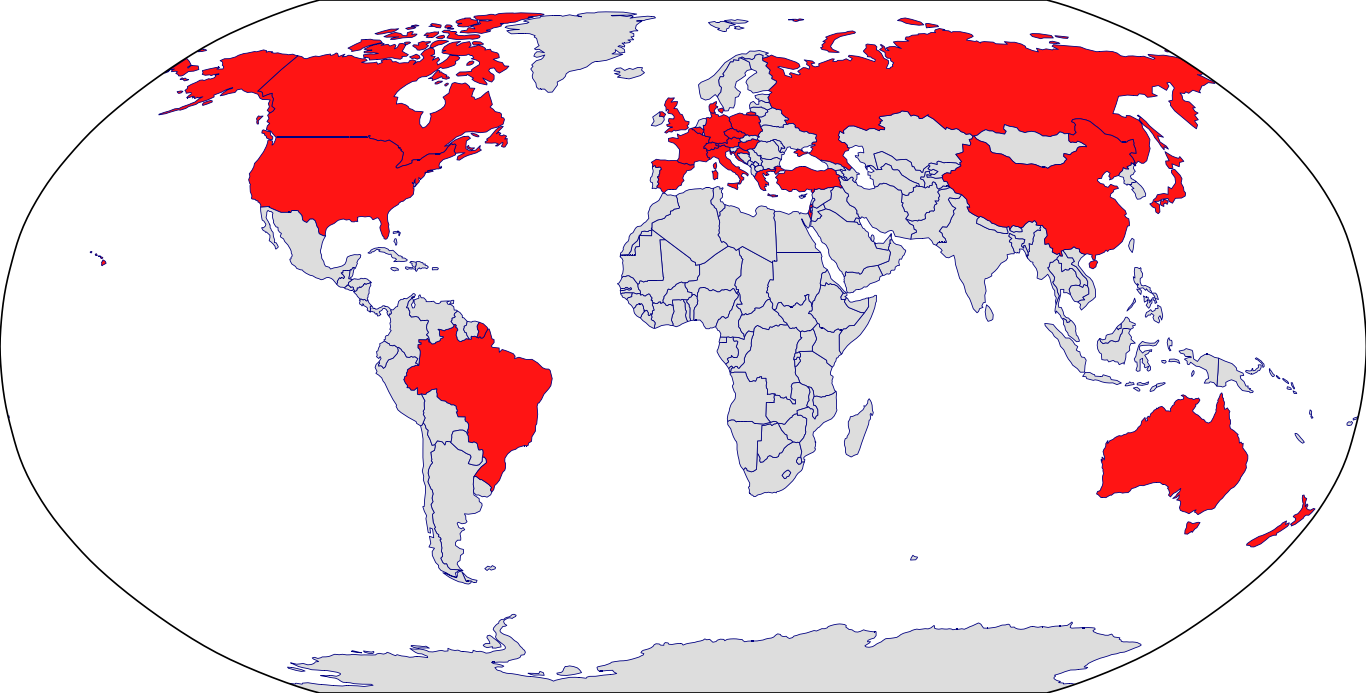
Countries in Clinic: Australia, Belgium, Brazil, China, France, Germany, Israel, Japan, Spain, United Kingdom, United States
Active Clinical Trial Count: 7
Recent & Upcoming Milestones
Highest Development Phases
Phase 3: Cytomegalovirus Infections|Hematopoietic Stem Cell Transplant|Transplantation Unspecified
Phase 1: Healthy Volunteers
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT06439342 |
TAK-620-3002 | P3 |
Recruiting |
Cytomegalovirus Infections |
2026-12-31 |
27% |
2025-03-01 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments |
jRCT2031230753 |
jRCT2031230753 | P3 |
Recruiting |
Transplantation Unspecified|Hematopoietic Stem Cell Transplant |
2026-11-22 |
21% |
||
NCT05319353 |
TAK-620-2004 | P3 |
Recruiting |
Cytomegalovirus Infections |
2027-01-18 |
21% |
2025-07-18 |
Primary Endpoints |
jRCT2021210056 |
jRCT2021210056 | P3 |
Completed |
Transplantation Unspecified|Hematopoietic Stem Cell Transplant |
2023-06-27 |
8% |
||
NCT05137717 |
TAK-620-3001 | P3 |
Completed |
Cytomegalovirus Infections |
2023-06-27 |
8% |
2024-06-06 |
|
2023-508988-73-00 |
TAK-620-2004 | P3 |
Temporary halt |
Transplantation Unspecified|Hematopoietic Stem Cell Transplant |
2027-03-31 |
21% |
2025-05-02 |
Treatments |
NCT05918822 |
TAK-620-1024 | P1 |
Completed |
Healthy Volunteers |
2023-08-25 |
69% |
2024-06-06 |
Primary Endpoints|Treatments |


