Product Description
Naperiglipron (LY3549492) is a small molecule (non-peptide), oral GLP-1 receptor agonist. (Sourced from: https://www.guidetopharmacology.org/GRAC/LigandDisplayForward?tab=clinical&ligandId=14175)
Mechanisms of Action: GLP-1 Agonist
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Company: Eli Lilly
Company Location: Eastern America
Company Founding Year: 1876
Additional Commercial Interests: None
Clinical Description
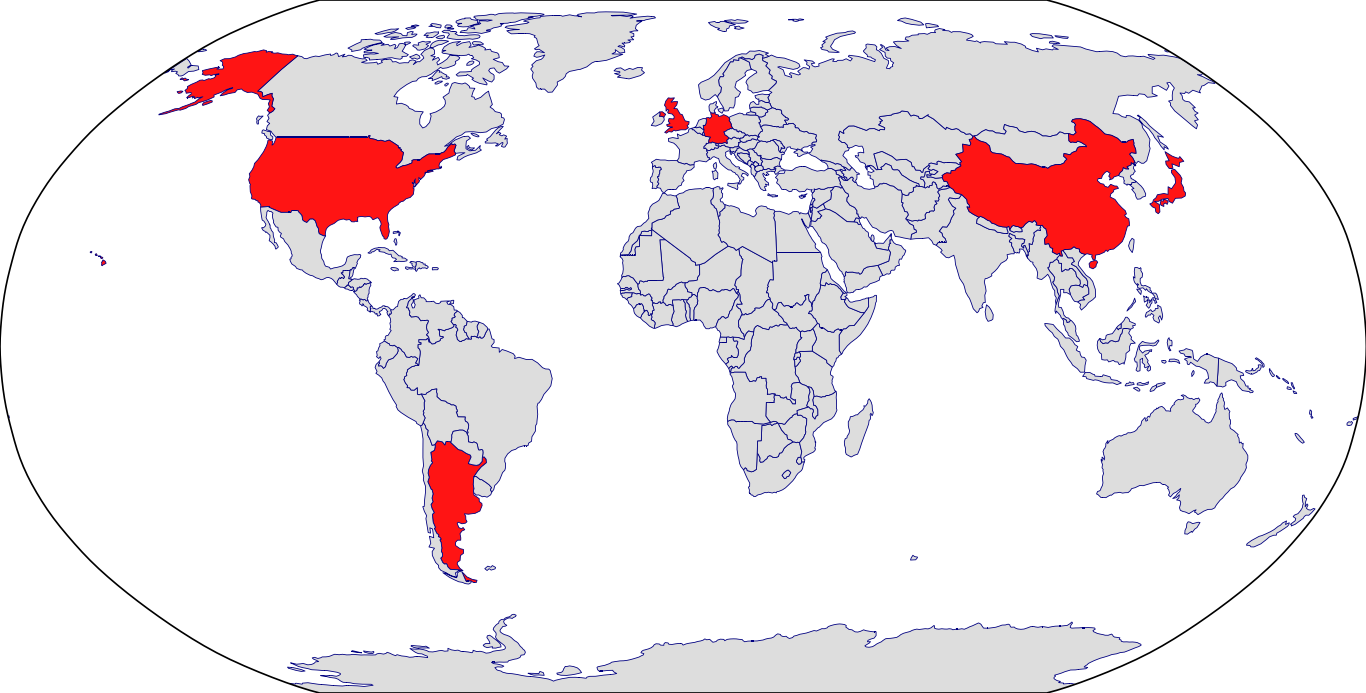
Countries in Clinic: Argentina, China, Germany, Japan, United Kingdom, United States
Active Clinical Trial Count: 9
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Obesity|Overweight
Phase 1: Glucose Metabolism Disorders|Healthy Volunteers|Type 2 Diabetes
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT06683508 |
W8M-MC-CWMM | P2 |
Active, not recruiting |
Overweight|Obesity |
2026-04-01 |
12% |
2025-04-23 |
Primary Endpoints|Treatments|Trial Status |
NCT06869018 |
J3H-JE-GZNG | P1 |
Active, not recruiting |
Type 2 Diabetes|Glucose Metabolism Disorders |
2026-03-01 |
88% |
2026-01-21 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments |
jRCT2071240136 |
jRCT2071240136 | P1 |
Recruiting |
Type 2 Diabetes |
2026-02-28 |
|||
NCT07073170 |
J3H-GH-GZNH | P1 |
Completed |
Type 2 Diabetes |
2026-01-27 |
50% |
2026-03-04 |
|
CTR20252656 |
CTR20252656 | P1 |
Completed |
Type 2 Diabetes |
2026-01-27 |
2026-02-10 |
Primary Completion Date|Study Completion Date|Treatments|Trial Status |
|
NCT05327595 |
J3H-MC-GZNB | P1 |
Completed |
Type 2 Diabetes |
2024-04-22 |
69% |
2024-05-31 |
Patient Enrollment|Primary Endpoints|Treatments|Trial Status |
NCT06194500 |
J3H-MC-GZNC | P1 |
Completed |
Healthy Volunteers |
2024-02-15 |
2024-03-29 |
||
NCT06143956 |
W8M-MC-CWMM | P2 |
Recruiting |
Overweight|Obesity |
2026-06-01 |
50% |
2025-10-16 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments |
NCT07232732 |
J3H-MC-GZNJ | P1 |
Recruiting |
Obesity|Overweight |
2026-07-01 |
88% |
2026-03-07 |
Patient Enrollment|Primary Endpoints|Treatments |
Recent News Events
Date |
Type |
Title |
|---|


