Product Description
JTX-8064 is a humanized IgG4 monoclonal antibody designed to specifically bind to Leukocyte Immunoglobulin Like Receptor B2 (LILRB2/ILT4) and block interactions with its ligands.
Mechanisms of Action: ILT4 Inhibitor
Novel Mechanism: Yes
Modality: Antibody
Route of Administration: N/A
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Jounce
Company Location:
Company Founding Year: 2013
Additional Commercial Interests: None
Clinical Description
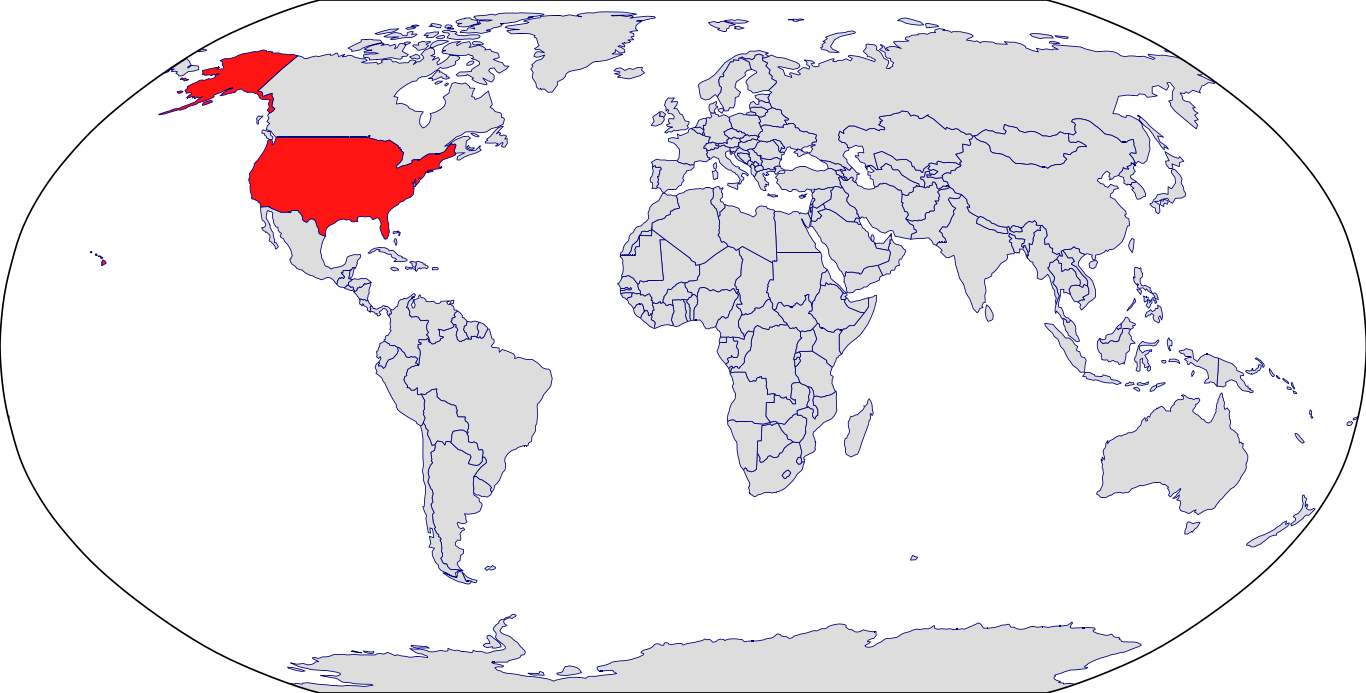
Countries in Clinic: United States
Active Clinical Trial Count: 1
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Oncology Solid Tumor Unspecified
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT04669899 |
JTX-8064-101 | P2 |
Completed |
Oncology Solid Tumor Unspecified |
2023-11-28 |
45% |
2024-04-10 |


