Product Description
Interferon-gamma (IFN-?) is a pleiotropic molecule with associated antiproliferative, pro-apoptotic and antitumor mechanisms.
Mechanisms of Action: IFNG Agonist
Novel Mechanism: No
Modality: Peptide/Protein
Route of Administration: Subcutaneous
FDA Designation: *
Approval Status: Approved
Approved Countries: Argentina | Australia | Austria | Bulgaria | Chile | Denmark | France | Germany | Greece | Hong Kong | Hungary | Ireland | Italy | Japan | Malta | Mexico | Netherlands | New Zealand | Norway | Philippines | Portugal | Russia | Saudi Arabia | Slovenia | Spain | Sweden | Switzerland | Ukraine | United Kingdom | United States | Vietnam
Approved Indications: None
Known Adverse Events: None
Company: InterMune
Company Location:
Company Founding Year: None
Additional Commercial Interests: None
Clinical Description
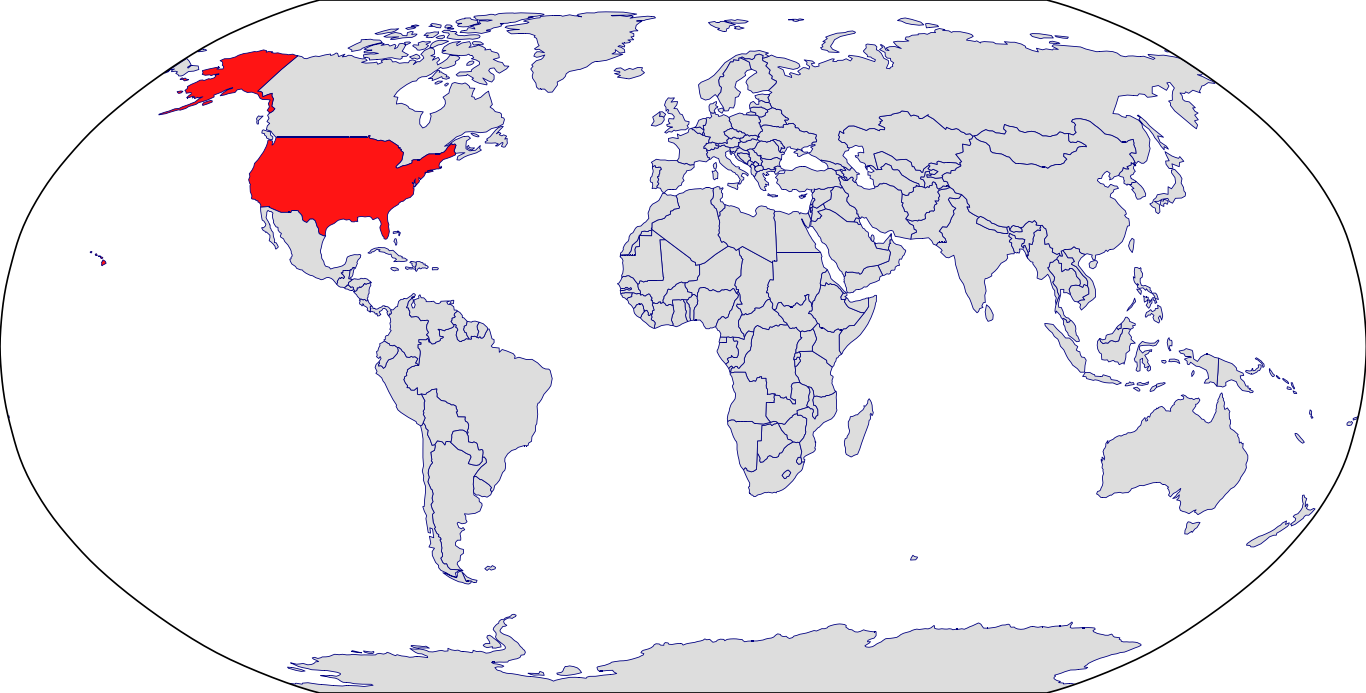
Countries in Clinic: United States
Active Clinical Trial Count: 1
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Acute Myeloid Leukemia|Myelodysplastic Syndrome|Preleukemia
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT06529731 |
alloSCT | P2 |
Recruiting |
Preleukemia|Acute Myeloid Leukemia|Myelodysplastic Syndrome |
2027-10-31 |
12% |
2024-11-01 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments |


