Product Description
INCAGN02385 is an Fc-engineered IgG1kappa antibody chosen for development based on its high-affinity binding to human LAG-3, cross-reactivity with cynomolgus monkey LAG-3, and ability to potently block LAG-3 binding with its MHC class II ligand. INCAGN02385 also enhances T cell responsiveness to TCR stimulation alone or in combination with PD-1/PD-L1 axis blockade. (Sourced from: https://aacrjournals.org/cancerres/article/78/13_Supplement/3819/628172/Abstract-3819-INCAGN02385-is-an-antagonist)
Mechanisms of Action: LAG3 Antagonist
Novel Mechanism: No
Modality: Antibody
Route of Administration: Intravenous
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Company: Incyte
Company Location: Eastern America
Company Founding Year: 1991
Additional Commercial Interests: None
Clinical Description
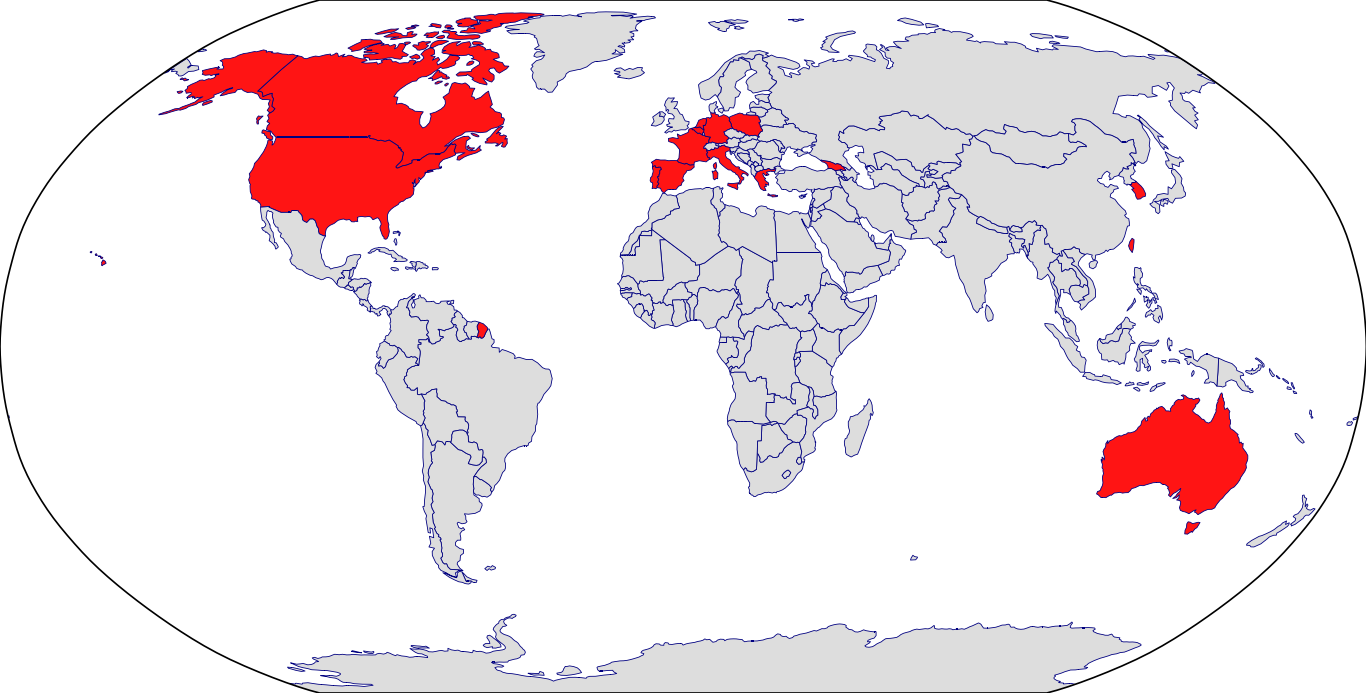
Countries in Clinic: Australia, Belgium, Canada, France, Georgia, Germany, Greece, Italy, Netherlands, Poland, Portugal, South Korea, Spain, Taiwan, United States
Active Clinical Trial Count: 4
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Carcinoma, Merkel Cell|Head and Neck Cancer|Melanoma|Squamous Cell Carcinoma
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT06056895 |
TRICK-MCC | P2 |
Active, not recruiting |
Carcinoma, Merkel Cell |
2027-01-31 |
12% |
2025-01-29 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
2023-504270-38-00 |
INCAGN 2385-203 | P2 |
Active, not recruiting |
Head and Neck Cancer|Squamous Cell Carcinoma |
2025-12-07 |
12% |
2025-05-02 |
Treatments |
NCT04370704 |
INCAGN 2385-201 | P2 |
Completed |
Melanoma |
2025-08-25 |
12% |
2025-10-04 |
Primary Endpoints |
NCT05287113 |
INCAGN 2385-203 | P2 |
Active, not recruiting |
Squamous Cell Carcinoma|Head and Neck Cancer |
2025-03-14 |
12% |
2025-08-27 |
Primary Endpoints|Study Completion Date |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
05/25/2023 |
News Article |
Data from Across Incyte's Oncology Portfolio Accepted for Presentation at the 2023 ASCO Annual Meeting and EHA2023 Hybrid Congress |
|
05/09/2023 |
News Article |
Agenus Provides Corporate Update and First Quarter 2023 Financial Results |


