Product Description
HRS-1167 is a drug candidate from Jiangsu HengRui Medicine for the treatment of Advanced Solid Tumors. (Sourced from: https://clinicaltrials.gov/ct2/show/NCT05473624?term=HRS-1167&draw=2&rank=1)
Mechanisms of Action: PARP Inhibitor
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Jiangsu Hengrui Medicine Co
Company Location: Asia Pacific
Company Founding Year: 1970
Additional Commercial Interests: None
Clinical Description
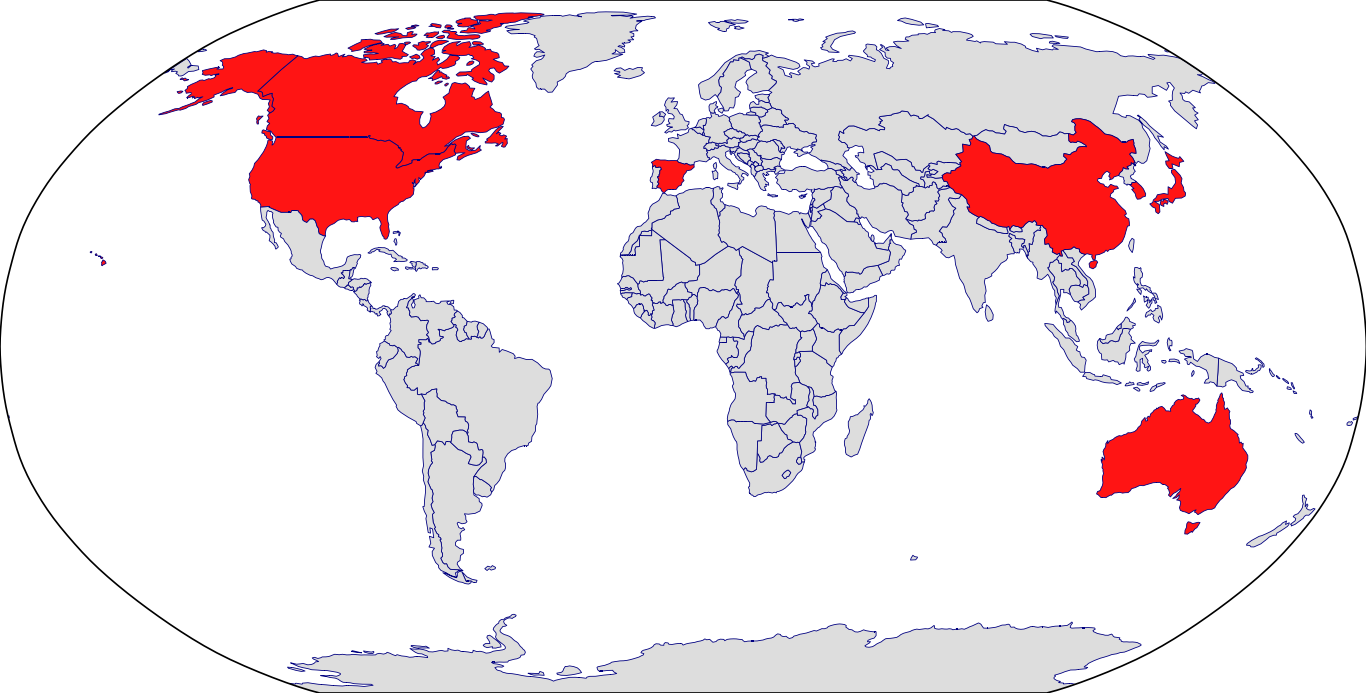
Countries in Clinic: Australia, Canada, China, Japan, Korea, South Korea, Spain, United Kingdom, United States
Active Clinical Trial Count: 9
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Prostate Cancer
Phase 1: Colorectal Cancer|Healthy Volunteers|Oncology Solid Tumor Unspecified|Ovarian Cancer|Small Cell Lung Cancer
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
jRCT2031240640 |
jRCT2031240640 | P1 |
Recruiting |
Small Cell Lung Cancer |
2027-10-06 |
|||
NCT06421935 |
DDriver 501 | P1 |
Active, not recruiting |
Ovarian Cancer |
2027-02-22 |
50% |
2025-12-20 |
Primary Endpoints|Treatments|Trial Status |
jRCT2031240418 |
jRCT2031240418 | P1 |
Recruiting |
Colorectal Cancer |
2026-04-07 |
|||
jRCT2031240255 |
jRCT2031240255 | P1 |
Recruiting |
Oncology Solid Tumor Unspecified |
2026-03-26 |
|||
NCT06689163 |
HRS-1167-201-A | P2 |
Recruiting |
Prostate Cancer |
2025-11-30 |
2% |
2025-01-03 |
Primary Endpoints|Start Date|Treatments|Trial Status |
CTR20234281 |
CTR20234281 | P1 |
Completed |
Healthy Volunteers|Oncology Solid Tumor Unspecified |
2024-02-05 |
2025-04-29 |
Patient Enrollment|Primary Completion Date|Start Date|Study Completion Date|Treatments|Trial Status |
|
NCT06198556 |
HRS-1167-102 | P1 |
Completed |
Oncology Solid Tumor Unspecified|Healthy Volunteers |
2024-02-05 |
12% |
2024-02-29 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
CTR20221773 |
CTR20221773 | P1 |
Recruiting |
Oncology Solid Tumor Unspecified |
None |
2025-04-29 |
||
NCT05473624 |
HRS-1167-I-101 | P1 |
Recruiting |
Oncology Solid Tumor Unspecified |
2026-12-31 |
50% |
2025-04-16 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |


