Product Description
HLX11 is a proposed biosimilar of pertuzumab (originator trade name: Perjeta®) (Sourced from: https://henlius.com/en/NewsDetails-2306-26.html)
Mechanisms of Action: EGFR2 Inhibitor
Novel Mechanism: Yes
Modality: Antibody
Route of Administration: Intravenous
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Henlius Biotech
Company Location: Asia Pacific
Company Founding Year: 2010
Additional Commercial Interests: None
Clinical Description
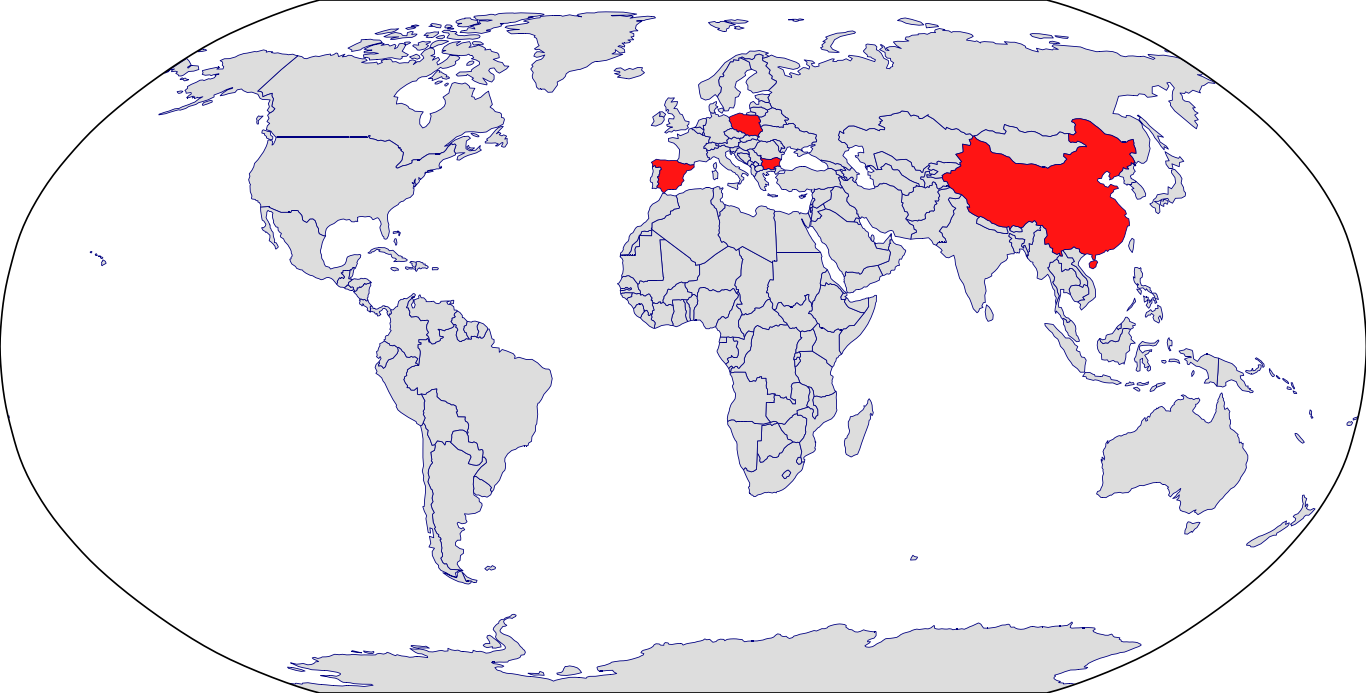
Countries in Clinic: Bulgaria, China, Poland, Spain
Active Clinical Trial Count: 2
Recent & Upcoming Milestones
Highest Development Phases
Phase 3: Breast Cancer
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
CTR20220391 |
CTR20220391 | P3 |
Completed |
Breast Cancer |
2025-09-30 |
2025-11-02 |
Primary Completion Date|Study Completion Date|Treatments|Trial Status |
|
NCT05346224 |
HLX11-BC301 | P3 |
Active, not recruiting |
Breast Cancer |
2024-05-15 |
35% |
2025-02-28 |
Primary Completion Date|Primary Endpoints |


