Product Description
An orally bioavailable inhibitor of nuclear enzyme poly(ADP-ribose) polymerase (PARP) 1, with potential antineoplastic activity. Upon oral administration, PARP1 inhibitor GS-0201 selectively binds to and inhibits the activity of PARP1, thereby preventing the repair of damaged DNA via the base excision repair (BER) pathway. This enhances the accumulation of DNA strand breaks and promotes genomic instability eventually leading to apoptosis. PARP1 catalyzes post-translational ADP-ribosylation of nuclear proteins that signal and recruit other proteins to repair damaged DNA and plays a key role in the repair of single strand DNA (ssDNA) breaks and double-strand breaks (DSBs). (Sourced from: https://www.cancer.gov/publications/dictionaries/cancer-drug/def/parp1-inhibitor-gs-0201)
Mechanisms of Action: PARP Inhibitor
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Gilead Sciences
Company Location: Western America
Company Founding Year: 1987
Additional Commercial Interests: None
Clinical Description
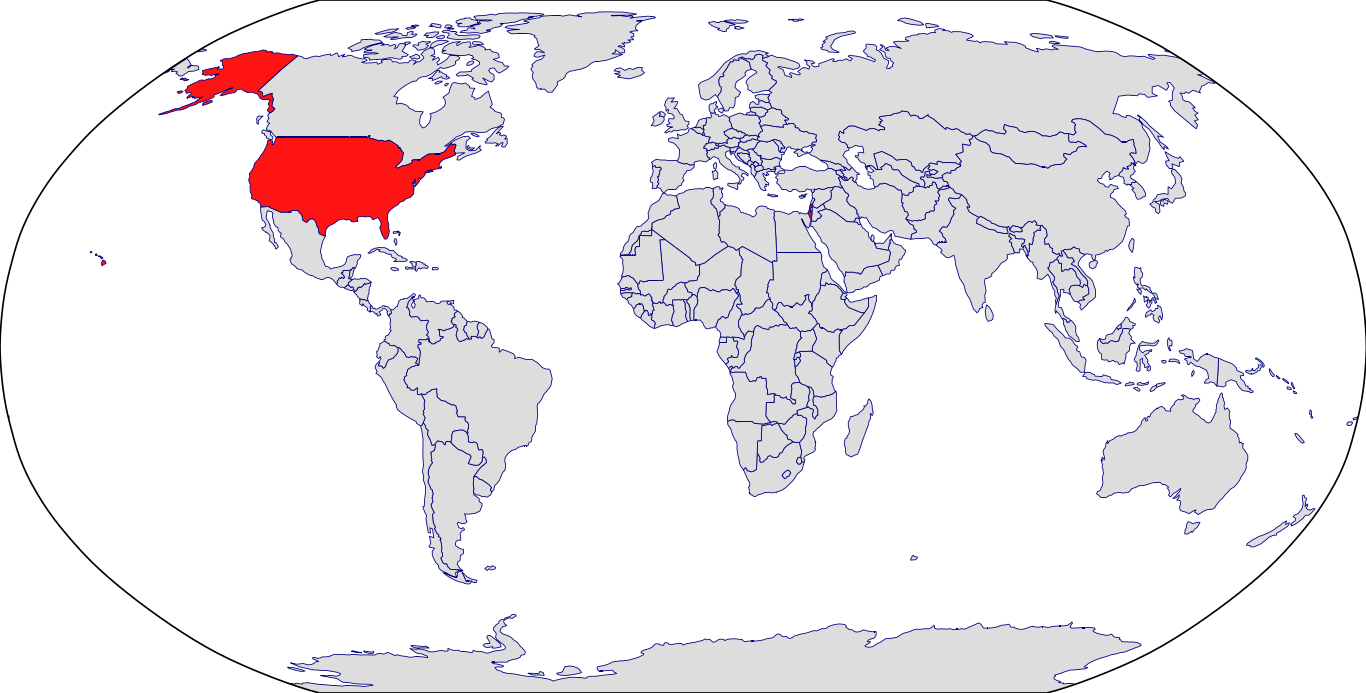
Countries in Clinic: Israel, United States
Active Clinical Trial Count: 1
Recent & Upcoming Milestones
Highest Development Phases
Phase 1: Oncology Solid Tumor Unspecified
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT06167317 |
GS-US-686-6854 | P1 |
Recruiting |
Oncology Solid Tumor Unspecified |
2028-09-01 |
12% |
2025-08-27 |
Recent News Events
Date |
Type |
Title |
|---|


