Product Description
SELLAS' lead product candidate, galinpepimut-S (GPS), targets malignancies and tumors characterized by an overexpression of the WT1 antigen. SELLAS' WT1 immunotherapy is comprised of four peptide chains, two of which are modified chains that induce a strong innate immune response (CD4+/CD8+) against the WT1 antigen and access a broad range of HLA types. When administered to a patient, GPS' induced immune response has the potential to recognize and destroy cancer cells and provide ongoing support and memory to the immune system so that it can continue to target and destroy recurring tumors and residual cancer cells. (Sourced from: https://www.sellaslifesciences.com/galinpepimut-s-gps-therapy/#section=therapy)
Mechanisms of Action: Vaccine, WT1
Novel Mechanism: Yes
Modality: Vaccine
Route of Administration: Subcutaneous, Oral
FDA Designation: *
Approval Status: Not Approved
Approved Countries: Australia | Chile | Colombia
Approved Indications: None
Known Adverse Events: None
Company: Sellas Life Sciences
Company Location:
Company Founding Year: None
Additional Commercial Interests: None
Clinical Description
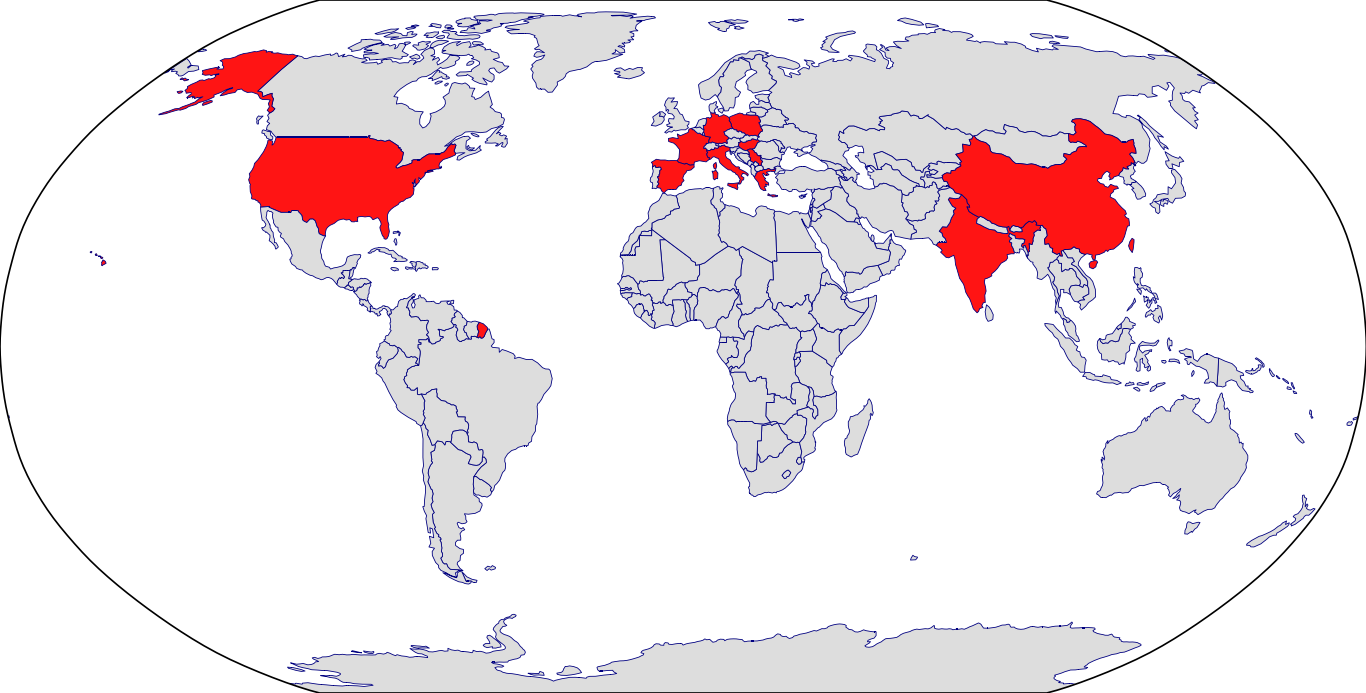
Countries in Clinic: China, France, Germany, Greece, Hungary, India, Italy, Poland, Serbia, Spain, Taiwan, United States
Active Clinical Trial Count: 4
Recent & Upcoming Milestones
- Clinical Outcomes Reported - Sellas Life Sciences presented P3 Acute Myeloid Leukemia results on 2025-12-29 for Galinpepimut-s
- Clinical Outcomes Reported - Sellas Life Sciences announced they will present P3 Acute Myeloid Leukemia results in 4Q25 for Galinpepimut-s
- Clinical Outcomes Reported - Sellas Life Sciences announced they will present P3 Acute Myeloid Leukemia results in YE25 for Galinpepimut-s
Highest Development Phases
Phase 3: Acute Myeloid Leukemia
Phase 1: Lymphoma, Non-Hodgkin|Multiple Myeloma|Myelodysplastic Syndrome|Oncology Hematological Unspecified|Oncology Unspecified
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05320809 |
3D189-CN-001 | P1 |
Active, not recruiting |
Lymphoma, Non-Hodgkin|Multiple Myeloma|Myelodysplastic Syndrome |
2026-06-01 |
50% |
2025-05-31 |
|
NCT04229979 |
REGAL | P3 |
Active, not recruiting |
Acute Myeloid Leukemia |
2025-12-01 |
33% |
2025-10-02 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Treatments |
CTR20221740 |
CTR20221740 | P1 |
Active, not recruiting |
Oncology Unspecified|Oncology Hematological Unspecified |
None |
2025-04-29 |
Patient Enrollment|Start Date|Treatments|Trial Status |
|
2024-516405-23-00 |
SLSG18-301 | P3 |
Active, not recruiting |
Acute Myeloid Leukemia |
2026-06-09 |
2025-05-02 |
Treatments |


