Product Description
An orally available, small molecule inhibitor of vascular endothelial growth factor receptors (VEGFRs), with potential anti-angiogenic and antineoplastic activities. (Sourced from: https://www.cancer.gov/publications/dictionaries/cancer-drug/def/fruquintinib#:~:text=An%20orally%20available%2C%20small%20molecule,anti%2Dangiogenic%20and%20antineoplastic%20activities.)
Mechanisms of Action: VEGFR Inhibitor
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: Fast Track - Colorectal Cancer|Oncology Solid Tumor UnspecifiedPriority Review - Endometrial Cancer *
Approval Status: Approved
Approved Countries: China
Approved Indications: None
Known Adverse Events: None
Company: Takeda
Company Location: Asia Pacific
Company Founding Year: 1781
Additional Commercial Interests: Hutchison Medipharma
Clinical Description
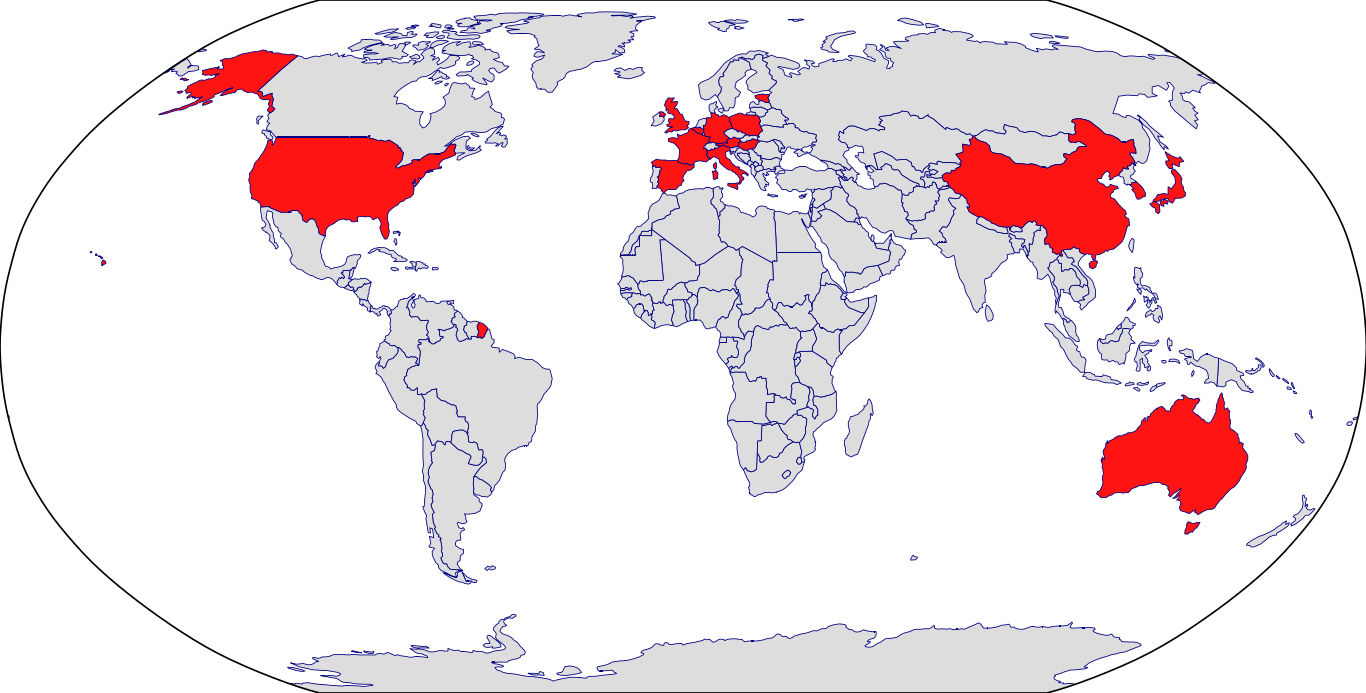
Countries in Clinic: Austria, China, France, Germany, Italy, Korea, South Korea, Spain, United Kingdom, United States, Unknown Location
Active Clinical Trial Count: 15
Recent & Upcoming Milestones
- Clinical Outcomes Reported - HUTCHMED presented P2 Renal Cell Carcinoma results on 2025-12-05 for Fruquintinib
- Clinical Outcomes Reported - HUTCHMED presented P3 Renal Cell Carcinoma results on 2025-09-11 for Fruquintinib
- Clinical Outcomes Reported - HUTCHMED presented P3 Oncology Solid Tumor Unspecified results on 2024-05-31 for Fruquintinib
Highest Development Phases
Phase 3: Adenocarcinoma|Colorectal Cancer|Endometrial Cancer|Gastrointestinal Cancer|Renal Cell Carcinoma
Phase 2: Acute Respiratory Distress Syndrome|Liver Cancer|Non-Small-Cell Lung Cancer|Oncology Solid Tumor Unspecified|Small Cell Lung Cancer
Phase 1: Kidney Diseases
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05216354 |
2021-013-00US2 | P1 |
Completed |
Kidney Diseases |
2023-06-26 |
23% |
2023-08-02 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
2024-519929-38-00 |
QUINTIS | P2 |
Not yet recruiting |
Liver Cancer|Colorectal Cancer |
2029-12-31 |
|||
NCT07136077 |
NCI-2025-06047 | P2 |
Recruiting |
Colorectal Cancer |
2026-04-01 |
12% |
2026-02-28 |
Primary Endpoints|Treatments|Trial Status |
NCT04716634 |
BGB-A317-fruquintinib-201 | P2 |
Completed |
Colorectal Cancer|Gastrointestinal Cancer|Non-Small-Cell Lung Cancer|Acute Respiratory Distress Syndrome|Small Cell Lung Cancer |
2024-02-22 |
42% |
2025-03-27 |
Primary Endpoints|Treatments |
CTR20211070 |
CTR20211070 | P2 |
Completed |
Oncology Solid Tumor Unspecified |
2024-02-22 |
42% |
2025-04-29 |
Patient Enrollment|Primary Completion Date|Start Date|Study Completion Date|Treatments|Trial Status |
NCT07270991 |
FRUQUITAS | P3 |
Recruiting |
Adenocarcinoma|Gastrointestinal Cancer |
2030-11-30 |
12% |
2026-02-06 |
Primary Endpoints|Start Date|Treatments|Trial Status |
NCT06584032 |
2024-013-00CH1 | P3 |
Recruiting |
Endometrial Cancer |
2029-01-08 |
21% |
2025-01-08 |
|
NCT05522231 |
FRUSICA-2 | P3 |
Active, not recruiting |
Renal Cell Carcinoma |
2025-01-01 |
28% |
2025-01-04 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Treatments|Trial Status |
CTR20181175 |
CTR20181175 | P1 |
Recruiting |
Non-Small-Cell Lung Cancer |
None |
2025-04-29 |
Patient Enrollment|Treatments |
|
CTR20182543 |
CTR20182543 | P1 |
Recruiting |
Colorectal Cancer |
None |
2025-04-29 |
||
NCT06992258 |
FRUITION | P2 |
Recruiting |
Colorectal Cancer |
2027-12-31 |
12% |
2025-12-23 |
Primary Endpoints|Start Date|Treatments|Trial Status |
NCT07011576 |
FRUITFUL | P2 |
Recruiting |
Colorectal Cancer |
2027-01-01 |
12% |
2025-08-13 |
Primary Endpoints|Start Date|Treatments|Trial Status |
NCT03903705 |
2018-013-00CH3 | P2 |
Completed |
Endometrial Cancer |
2023-11-15 |
22% |
2025-04-08 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
jRCT2041200099 |
jRCT2041200099 | P3 |
Active, not recruiting |
Colorectal Cancer |
2022-10-31 |
|||
NCT07042685 |
PRO00039815 (HMCC-GI25-001) | P2 |
Recruiting |
Colorectal Cancer |
2028-12-01 |
12% |
2026-01-01 |
Primary Endpoints|Start Date |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
02/02/2026 |
News Article |
FRUZAQLA(TM) (fruquintinib capsules) now reimbursed in Ontario to treat metastatic colorectal cancer (mCRC) |
|
01/23/2026 |
News Article |
Adagene Provides Business Update and 2026 Objectives |
|
01/08/2026 |
News Article |
KAHR Bio Announces Strong Topline Phase 2 Results for DSP107 in Combination with Anti-PD-L1 Therapy in Colorectal Cancer |
|
12/25/2025 |
News Article |
China's First Domestic Anti-CTLA-4 Monoclonal Antibody, Innovent's TABOSUN® (Ipilimumab N01 Injection) Received NMPA Approval |


