Product Description
Ethambutol is a medication used in the management and treatment of tuberculosis. It is a bacteriostatic drug that inhibits cell wall synthesis. (Sourced from: https://www.ncbi.nlm.nih.gov/books/NBK559050/)
Mechanisms of Action: Arabinosyl Transferase Inhibitor, Cell Wall Synthesis Inhibitor
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Approved
Approved Countries: Australia | Austria | Bangladesh | Belgium | Brazil | Bulgaria | Canada | Chile | China | Colombia | Czech | Denmark | Dominican Republic | Egypt | Finland | France | Germany | Greece | Hong Kong | Hungary | India | Indonesia | Ireland | Italy | Japan | Korea | Latvia | Lithuania | Luxembourg | Malaysia | Malta | Morocco | Netherlands | New Zealand | Pakistan | Peru | Philippines | Poland | Portugal | Romania | Russia | Singapore | Slovakia | Slovenia | South Africa | Spain | Sweden | Switzerland | Taiwan | Thailand | Turkey | Ukraine | United Kingdom | United States | Uruguay | Venezuela | Vietnam
Approved Indications: None
Known Adverse Events: None
Company: Insmed
Company Location: Eastern America
Company Founding Year: 1999
Additional Commercial Interests: None
Clinical Description
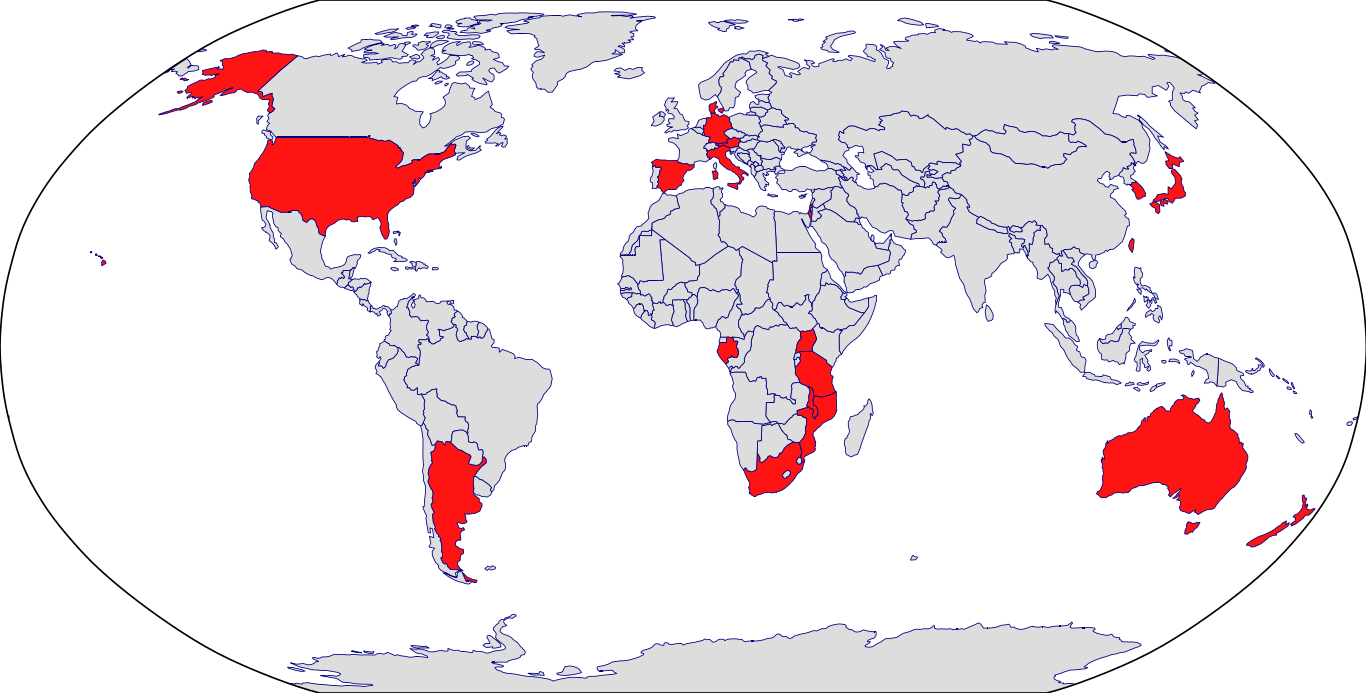
Countries in Clinic: Argentina, Australia, Austria, Denmark, Gabon, Germany, Israel, Italy, Japan, Korea, Malawi, Mozambique, New Zealand, South Africa, South Korea, Spain, Taiwan, Tanzania, Uganda, United States
Active Clinical Trial Count: 5
Recent & Upcoming Milestones
Highest Development Phases
Phase 3: Communicable Diseases|Mycobacterium Infections, Nontuberculous|Mycobacterium avium-intracellulare Infection|Nontuberculous Mycobacterial Lung Disease|Other
Phase 2: Tuberculosis, Pulmonary
Phase 1: Healthy Volunteers
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05807399 |
PanACEA - STEP2C -01 | P2 |
Recruiting |
Tuberculosis, Pulmonary |
2027-10-31 |
50% |
2025-10-08 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Study Completion Date |
NCT04630145 |
MAC-LD | P3 |
Completed |
Other |
2024-11-06 |
39% |
2025-12-23 |
Primary Endpoints|Study Completion Date|Treatments|Trial Status |
2020-002545-42 |
ARISE | P3 |
Completed |
Nontuberculous Mycobacterial Lung Disease |
2023-05-09 |
17% |
2025-05-06 |
Primary Completion Date|Study Completion Date|Treatments|Trial Status |
NCT04677543 |
ARISE | P3 |
Completed |
Mycobacterium Infections, Nontuberculous|Mycobacterium avium-intracellulare Infection|Communicable Diseases |
2023-05-09 |
17% |
2024-06-28 |
Primary Endpoints |
NCT05966688 |
SPR720-102 | P1 |
Completed |
Healthy Volunteers |
2024-01-30 |
69% |
2024-03-05 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments |


