Product Description
Escient Pharmaceuticals is developing EP-547, an MRGPRX4-targeted oral therapy, as a treatment for cholestatic and uremic pruritus. (Sourced from: https://www.escientpharma.com/escient-pharmaceuticals-announces-positive-results-from-phase-1-study-of-ep547-an-mrgprx4-targeted-oral-therapy-for-cholestatic-and-uremic-pruritus/)
Mechanisms of Action: MRGPRX4 Antagonist
Novel Mechanism: Yes
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Company: Escient Pharmaceuticals, Inc
Company Location:
Company Founding Year: None
Additional Commercial Interests: None
Clinical Description
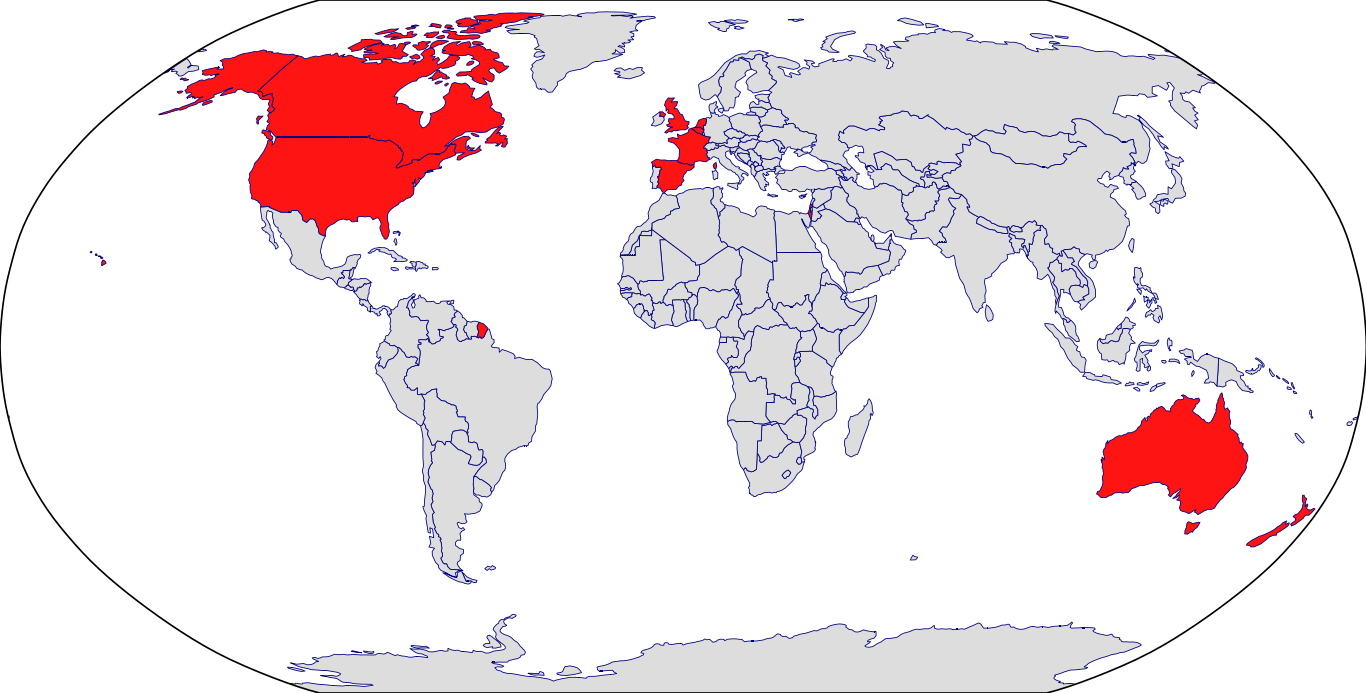
Countries in Clinic: Belgium, Canada, France, Israel, Netherlands, Spain, United Kingdom, United States
Active Clinical Trial Count: 2
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Biliary Cirrhosis|Cholangitis, Sclerosing|Liver Cirrhosis|Pruritus
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05525520 |
PACIFIC | P2 |
Completed |
Pruritus|Cholangitis, Sclerosing|Biliary Cirrhosis|Liver Cirrhosis |
2024-07-17 |
12% |
2024-10-16 |
Primary Completion Date|Primary Endpoints|Study Completion Date |
2021-002526-25 |
PACIFIC | P2 |
Completed |
Cholangitis, Sclerosing|Pruritus|Biliary Cirrhosis |
2024-09-05 |
2025-06-27 |
Treatments |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
07/30/2024 |
News Article |
Incyte Reports 2024 Second Quarter Financial Results and Provides Updates on Key Clinical Programs |
|
06/12/2024 |
News Article |
Science 37 Surpasses Enrollment Goals, Accelerates Phase 2 Rare Disease Clinical Trial |
|
05/30/2024 |
News Article |
Incyte Completes Acquisition of Escient Pharmaceuticals |
|
04/30/2024 |
News Article |
Incyte Reports 2024 First Quarter Financial Results and Provides Updates on Key Clinical Programs |


