Product Description
Duloxetine is an antidepressant medication that works in the brain. It is approved for the treatment of major depressive disorder (MDD), generalized anxiety disorder (GAD), diabetic peripheral neuropathic pain (DPNP), fibromyalgia, and chronic musculoskeletal pain. (Sourced from: https://www.nami.org/About-Mental-Illness/Treatments/Mental-Health-Medications/Types-of-Medication/Duloxetine-(Cymbalta))
Mechanisms of Action: SSR Inhibitor, NRI Inhibitor
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Approved
Approved Countries: Algeria | Argentina | Australia | Austria | Bangladesh | Belgium | Bosnia | Brazil | Bulgaria | Canada | Chile | China | Colombia | Croatia | Cyprus | Czech | Denmark | Dominican Republic | Ecuador | Egypt | Estonia | European Medicines Agency | Finland | France | Germany | Greece | Hong Kong | Hungary | Iceland | India | Indonesia | Ireland | Israel | Italy | Japan | Jordan | Korea | Latvia | Lebanon | Lithuania | Luxembourg | Malaysia | Malta | Mexico | Morocco | Netherlands | New Zealand | Norway | Pakistan | Peru | Philippines | Poland | Portugal | Romania | Russia | Saudi Arabia | Serbia | Singapore | Slovakia | Slovenia | South Africa | Spain | Sri Lanka | Sweden | Switzerland | Taiwan | Thailand | Tunisia | Turkey | Ukraine | United Arab Emirates | United Kingdom | United States | Uruguay | Venezuela | Vietnam
Approved Indications: None
Known Adverse Events: None
Company: Eli Lilly
Company Location: Eastern America
Company Founding Year: 1876
Additional Commercial Interests: None
Clinical Description
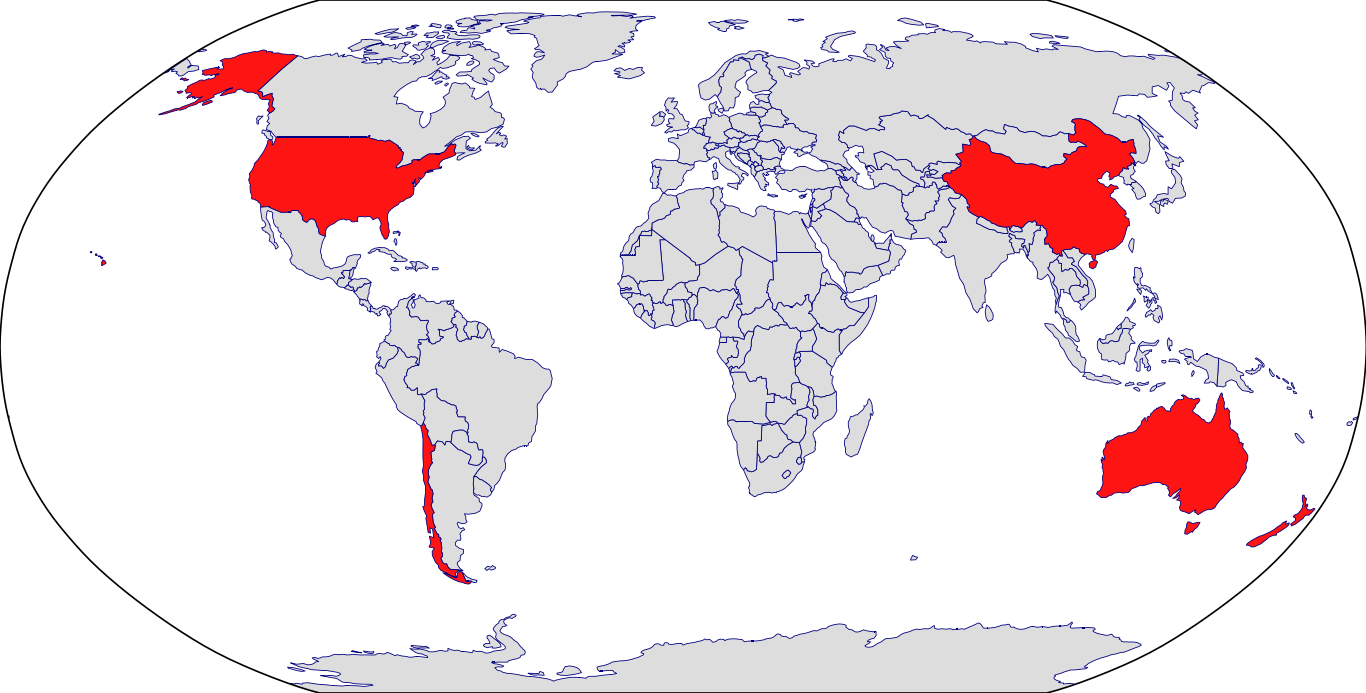
Countries in Clinic: Australia, China, Hong Kong, New Zealand, United States
Active Clinical Trial Count: 5
Recent & Upcoming Milestones
Highest Development Phases
Phase 3: Depressive Disorder|Depressive Disorder, Major|Osteoarthritis, Knee
Phase 1: Dermatitis, Atopic
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
ACTRN12619000395134p |
2006-7041-83/hah | P1 |
Not yet recruiting |
Dermatitis, Atopic |
2019-07-15 |
|||
CTR20251261 |
CTR20251261 | P3 |
Completed |
Depressive Disorder, Major |
2026-02-04 |
2026-02-15 |
Primary Completion Date|Study Completion Date|Treatments|Trial Status |
|
CTR20243567 |
CTR20243567 | P3 |
Completed |
Depressive Disorder |
2025-07-22 |
2025-11-09 |
Patient Enrollment|Primary Completion Date|Start Date|Study Completion Date|Treatments|Trial Status |
|
NCT04504812 |
SKOAP | P3 |
Completed |
Osteoarthritis, Knee |
2025-04-08 |
2025-04-19 |
Primary Endpoints |
|
CTR20180202 |
CTR20180202 | P1 |
Active, not recruiting |
Depressive Disorder |
None |
2025-04-29 |


