Product Description
Ionis is developing Donidalorsen as a treatment for Hereditary angioedema (HAE). (Sourced from: https://www.ionispharma.com/medicines/ionis-pkk-l/)
Mechanisms of Action: Gene Therapy, PKK
Novel Mechanism: No
Modality: Gene Therapy
Route of Administration: Subcutaneous
FDA Designation: *
Approval Status: Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Ionis
Company Location: Western America
Company Founding Year: 1989
Additional Commercial Interests: Otsuka
Clinical Description
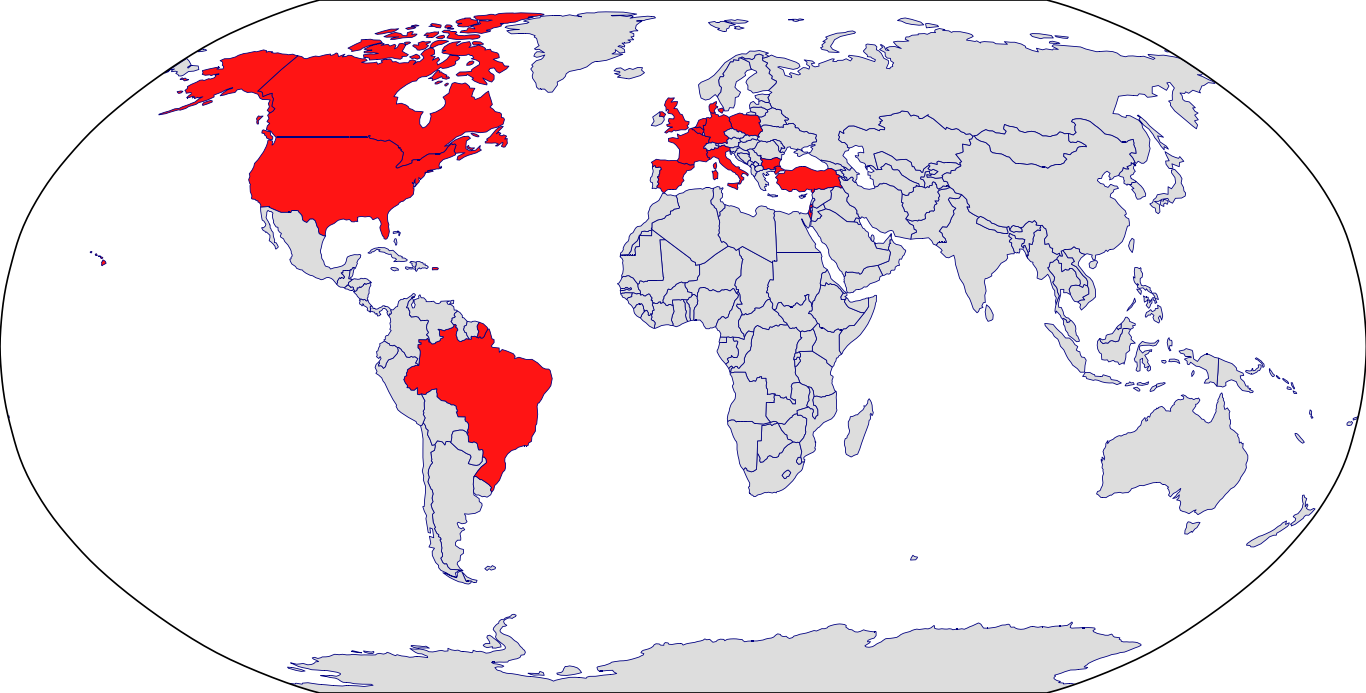
Countries in Clinic: Belgium, Bulgaria, Canada, Denmark, France, Germany, Israel, Italy, Netherlands, Poland, Puerto Rico, Spain, Turkey, United Kingdom, United States
Active Clinical Trial Count: 8
Recent & Upcoming Milestones
- Clinical Outcomes Reported - Ionis presented P3 Angioedema results on 2026-02-27 for Donidalorsen
- Clinical Outcomes Reported - Ionis presented P2 Angioedemas, Hereditary results on 2025-11-06 for Donidalorsen
- Clinical Outcomes Reported - Ionis presented P3 Angioedemas, Hereditary results on 2025-07-21 for Donidalorsen
Highest Development Phases
Phase 3: Angioedemas, Hereditary
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05392114 |
ISIS 721744-CS7 | P3 |
Active, not recruiting |
Angioedemas, Hereditary |
2026-12-01 |
21% |
2025-06-25 |
Patient Enrollment|Primary Endpoints|Treatments|Trial Status |
2024-517249-15-00 |
ISIS721744-CS3 | P2 |
Completed |
Angioedemas, Hereditary |
2024-12-19 |
2025-05-02 |
Treatments |
|
2025-523499-22-00 |
ISIS 721744-CS8 | P3 |
Not yet recruiting |
Angioedemas, Hereditary |
2029-06-30 |
23% |
||
NCT07298447 |
ISIS 721744-CS8 | P3 |
Not yet recruiting |
Angioedemas, Hereditary |
2029-06-01 |
23% |
2025-12-24 |
Primary Endpoints|Treatments |
2023-509201-77-00 |
ISIS 721744-CS7 | P3 |
Active, not recruiting |
Angioedemas, Hereditary |
2026-11-06 |
2025-05-02 |
Treatments |
|
2021-002571-19 |
2021-002571-19 | P3 |
Completed |
Angioedemas, Hereditary |
2023-11-09 |
25% |
2025-05-06 |
Primary Completion Date|Study Completion Date|Treatments|Trial Status |
NCT04307381 |
ISIS 721744-CS3 | P2 |
Active, not recruiting |
Angioedemas, Hereditary |
2025-04-01 |
12% |
2025-03-27 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
NCT05139810 |
OASIS-HAE | P3 |
Completed |
Angioedemas, Hereditary |
2023-11-09 |
25% |
2025-03-07 |
Primary Completion Date|Primary Endpoints |


