Product Description
Diclofenac is a nonsteroidal anti-inflammatory drug (NSAID) used to treat mild-to-moderate pain, and helps to relieve symptoms of arthritis (eg, osteoarthritis or rheumatoid arthritis), such as inflammation, swelling, stiffness, and joint pain. This medicine does not cure arthritis and will only help you as long as you continue to take it. (Sourced from: https://www.mayoclinic.org/drugs-supplements/diclofenac-oral-route/description/drg-20069748)
Mechanisms of Action: COX2 Inhibitor, COX1 Inhibitor
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral, Topical, Transdermal
FDA Designation: *
Approval Status: Approved
Approved Countries: Algeria | Argentina | Australia | Austria | Bangladesh | Belgium | Bosnia | Brazil | Bulgaria | Canada | Chile | China | Colombia | Croatia | Cyprus | Czech | Denmark | Dominican Republic | Ecuador | Egypt | Estonia | Finland | France | Germany | Greece | Hong Kong | Hungary | Iceland | India | Indonesia | Ireland | Israel | Italy | Japan | Jordan | Korea | Latvia | Lebanon | Lithuania | Luxembourg | Malaysia | Malta | Mexico | Morocco | Netherlands | New Zealand | Norway | Pakistan | Peru | Philippines | Poland | Portugal | Romania | Russia | Saudi Arabia | Serbia | Singapore | Slovakia | Slovenia | South Africa | Spain | Sri Lanka | Sweden | Switzerland | Taiwan | Thailand | Tunisia | Turkey | Ukraine | United Arab Emirates | United Kingdom | United States | Uruguay | Venezuela | Vietnam
Approved Indications: None
Known Adverse Events: None
Company: Novartis
Company Location: Europe
Company Founding Year: 1996
Additional Commercial Interests: None
Clinical Description
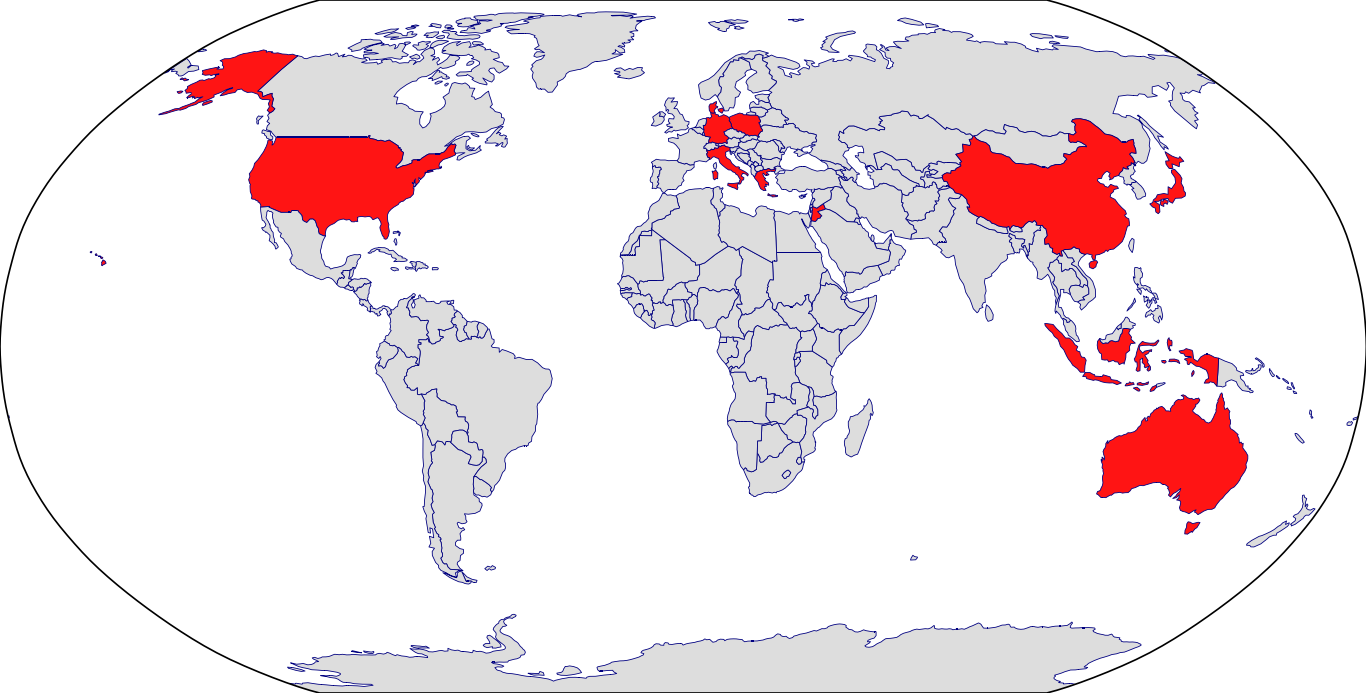
Countries in Clinic: Australia, China, Czech Republic, Denmark, Germany, Greece, Italy, Japan, Poland, United States
Active Clinical Trial Count: 24
Recent & Upcoming Milestones
Highest Development Phases
Phase 3: Acute Pain|Cancer Pain|Chronic Pain|Keratosis, Actinic|Low Back Pain|Osteoarthritis, Knee
Phase 2: Musculoskeletal Pain
Phase 1: Healthy Volunteers|Osteoarthritis|Pain Unspecified
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05254574 |
OA-01-2022 | P1 |
Recruiting |
Osteoarthritis |
2026-05-01 |
50% |
2025-02-28 |
Primary Completion Date|Primary Endpoints |
NCT05883241 |
BF-01-2023 | P1 |
Recruiting |
Pain Unspecified |
2026-02-01 |
50% |
2025-03-01 |
Primary Endpoints |
NCT06257537 |
KP-01-2024 | P2 |
Recruiting |
Osteoarthritis, Knee |
2026-02-01 |
50% |
2025-02-13 |
Primary Endpoints |
ACTRN12623000273684 |
ACTRN12623000273684 | P2 |
Completed |
Musculoskeletal Pain|Osteoarthritis, Knee |
2024-12-03 |
2026-02-15 |
Treatments |
|
2025-521578-32-00 |
DI-OR-PA | P3 |
Not yet recruiting |
Low Back Pain |
2027-03-31 |
|||
2024-518753-42-00 |
EPDICTIO75+4F01-2022 | P3 |
Active, not recruiting |
Chronic Pain|Acute Pain|Low Back Pain |
2025-03-31 |
2025-05-02 |
Treatments |
|
2019-003678-16 |
5%-ige KOH-Lösung vs. Placebo und Diclofenac-Gel zur Behandlung der Aktinischen Keratose | P3 |
Active, not recruiting |
Keratosis, Actinic |
2023-05-15 |
2022-03-13 |
Treatments |
|
NCT05752526 |
DARE-PDM1 | P1 |
Completed |
Pain Unspecified |
2023-10-10 |
12% |
2025-09-05 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Study Completion Date |
ACTRN12622001391763 |
ACTRN12622001391763 | P1 |
Completed |
Osteoarthritis |
2023-01-18 |
2026-02-15 |
Treatments |
|
NCT04640675 |
HP-5000-US-07 | P3 |
Not yet recruiting |
Osteoarthritis, Knee |
2022-08-01 |
15% |
2020-11-30 |
Primary Endpoints|Treatments |
JapicCTI-205103 |
JapicCTI-205103 | P3 |
Active |
Low Back Pain |
2021-07-31 |
|||
NCT05882812 |
KP-01-2022 | P2 |
Completed |
Osteoarthritis, Knee |
2023-12-31 |
50% |
2024-02-29 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
JapicCTI-205291 |
JapicCTI-205291 | P3 |
Planned |
Low Back Pain |
2020-10-31 |
|||
2015-004102-42 |
Diclofenac 2% in improvement of pain symptoms | P3 |
Active, not recruiting |
Acute Pain |
2017-11-28 |
2022-03-13 |
Treatments |
|
JapicCTI-173651 |
JapicCTI-173651 | P3 |
Active |
Cancer Pain |
None |
|||
NCT06693648 |
AMZ001 | P3 |
Active, not recruiting |
Osteoarthritis, Knee |
2025-12-22 |
10% |
2026-01-17 |
Primary Completion Date|Primary Endpoints |
2024-517404-11-00 |
AMZ001-007 | P3 |
Not yet recruiting |
Osteoarthritis, Knee |
2025-09-15 |
2025-05-01 |
Treatments |
|
2024-512786-13-00 |
DITH/VER | P3 |
Recruiting |
Low Back Pain|Acute Pain |
2025-01-02 |
2025-05-02 |
Treatments |
|
2021-006465-38 |
2021-006465-38 | P3 |
Completed |
Acute Pain |
2023-10-31 |
2025-07-09 |
Treatments |
|
NCT06729073 |
AMZ001 | P1 |
Completed |
Healthy Volunteers |
2025-06-10 |
88% |
2026-01-07 |
Primary Endpoints|Study Completion Date |
CTR20250470 |
CTR20250470 | P1 |
Completed |
Healthy Volunteers|Pain Unspecified |
2025-03-21 |
2025-05-04 |
Primary Completion Date|Start Date|Study Completion Date|Treatments|Trial Status |
|
ACTRN12624000341527 |
ACTRN12624000341527 | P1 |
Recruiting |
Osteoarthritis, Knee |
2024-07-14 |
2026-02-15 |
Treatments |
|
NCT05968482 |
AMZ001-008 | P1 |
Completed |
Healthy Volunteers |
2023-12-27 |
12% |
2025-04-10 |
Primary Endpoints|Study Completion Date |
ACTRN12623000261617 |
ACTRN12623000261617 | P1 |
Completed |
Osteoarthritis, Knee|Musculoskeletal Pain |
2023-11-17 |
2026-02-15 |
Treatments |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
03/11/2026 |
News Article |
Haleon Strengthens Commitment to China as Consumers Focus on Better Everyday Health |
|
03/09/2026 |
News Article |
Study in Nature Medicine Finds Taking Centrum Silver Daily May Slow Biological Aging |
|
02/10/2026 |
News Article |
New Sensodyne Clinical Repair Toothpaste Launches with Breakthrough Formula for Rapid, Clinically Proven Sensitivity Relief |
|
01/09/2026 |
News Article |
No serious adverse events to date in ongoing phase 1/2a clinical study of SYN321 |


