Product Description
In patients with chronic HFrEF, the haemodynamic effects of cimlanod and NTG are similar. The effects of cimlanod may be explained by venodilatation and preload reduction without additional inotropic or lusitropic effects. Ongoing trials of cimlanod will further define its potential role in the treatment of heart failure. (Sourced from: https://pubmed.ncbi.nlm.nih.gov/33620131/)
Mechanisms of Action: Unknown
Novel Mechanism: Yes
Modality: Small Molecule
Route of Administration: Intravenous
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Bristol-Myers Squibb
Company Location: Eastern America
Company Founding Year: 1989
Additional Commercial Interests: None
Clinical Description
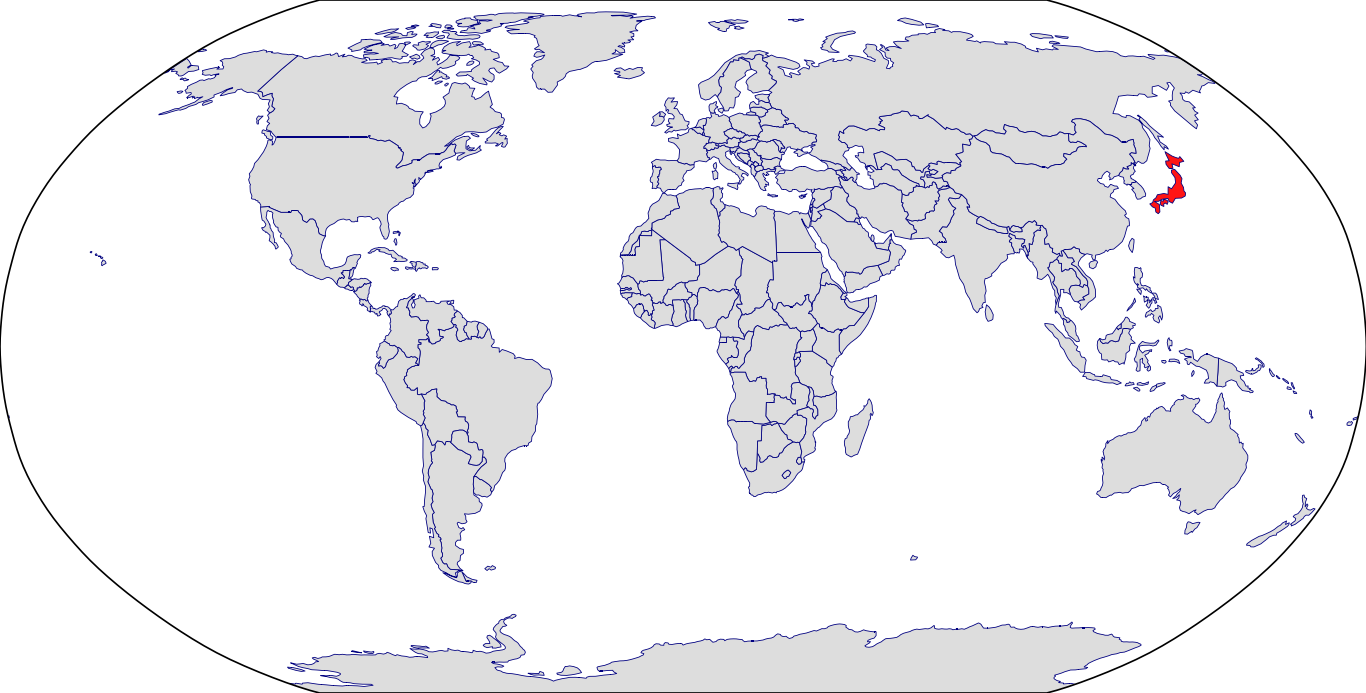
Countries in Clinic: Japan
Active Clinical Trial Count: 2
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Heart Failure, Acute|Heart Failure, Chronic
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
JapicCTI-184096 |
JapicCTI-184096 | P2 |
Active |
Heart Failure, Acute |
2019-08-01 |
|||
JapicCTI-173794 |
JapicCTI-173794 | P2 |
Active |
Heart Failure, Chronic |
2019-03-01 |


