Product Description
Canakinumab (ACZ885, Ilaris) is a human anti-IL-1beta monoclonal antibody developed by Novartis. its mode of action is based on the neutralization of 1beta signaling, resulting in suppression of inflammation in patients with disorders of autoimmune origin. (Sourced from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2828573/)
Mechanisms of Action: IL1B Inhibitor
Novel Mechanism: No
Modality: Antibody
Route of Administration: Subcutaneous
FDA Designation: *
Approval Status: Approved
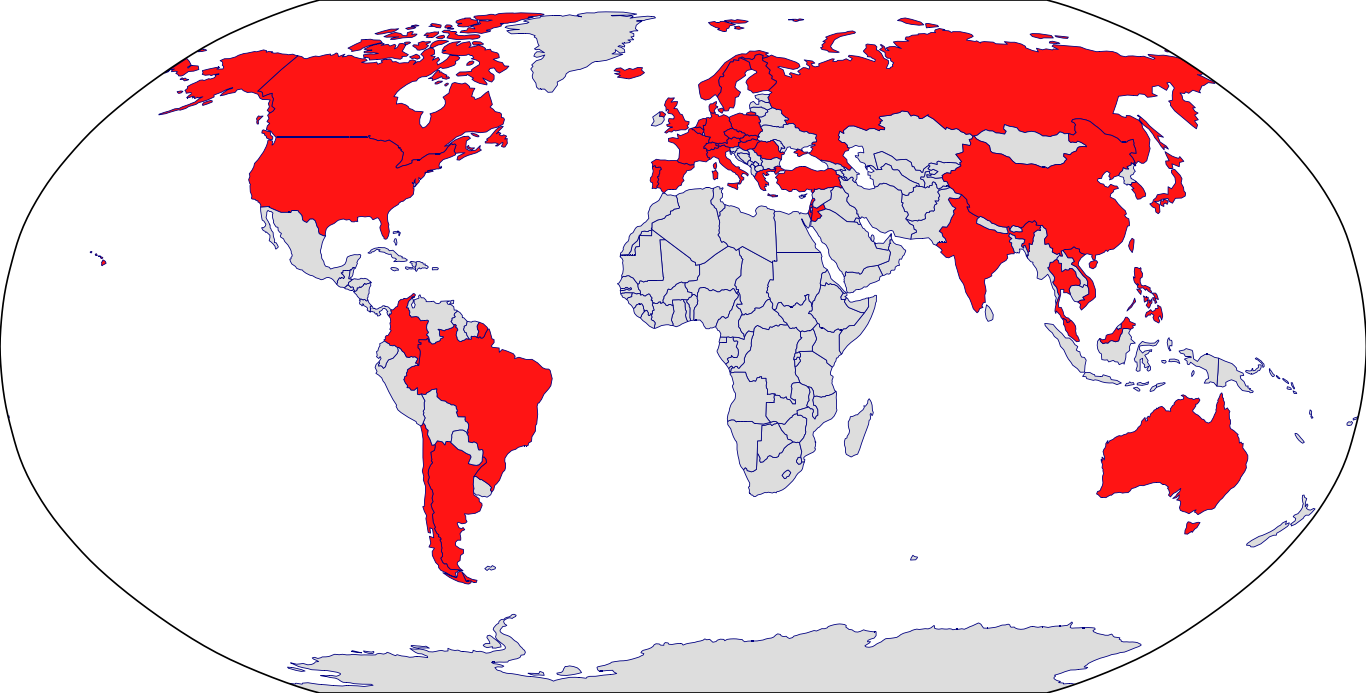
Approved Countries: Argentina | Australia | Austria | Belgium | Brazil | Canada | Chile | Colombia | Croatia | Cyprus | Czech | Denmark | Dominican Republic | Estonia | European Medicines Agency | Finland | France | Germany | Greece | Hong Kong | Hungary | Iceland | Ireland | Israel | Italy | Japan | Korea | Latvia | Lithuania | Luxembourg | Netherlands | New Zealand | Norway | Peru | Poland | Portugal | Romania | Russia | Saudi Arabia | Serbia | Singapore | Slovakia | Slovenia | South Africa | Spain | Sweden | Switzerland | Taiwan | Turkey | Ukraine | United Arab Emirates | United Kingdom | United States | Venezuela
Approved Indications: None
Company: Novartis
Company Location: Europe
Company Founding Year: 1996
Additional Commercial Interests: None
Clinical Description
Countries in Clinic: Germany, Japan, Russia, Spain, United States
Active Clinical Trial Count: 15
Recent & Upcoming Milestones
Highest Development Phases
Phase 3: Adenocarcinoma|COVID-19|Cold Urticaria|Cryopyrin-Associated Periodic Syndromes|Lung Cancer|Non-Small-Cell Lung Cancer|Pancreatic Cancer|Still's Disease, Adult-Onset
Phase 2: Acute Monocytic Leukemia|Acute Myelomonocytic Leukemia|Chronic Myelomonocytic Leukemia|Familial Mediterranean Fever|Juvenile Myelomonocytic Leukemia,|Myelodysplastic Syndrome|Preleukemia
Phase 1: Renal Cell Carcinoma
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT04028245 |
SPARC-1 | P1 |
Active, not recruiting |
Renal Cell Carcinoma |
2025-03-14 |
12% |
2025-08-08 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
NCT04239157 |
NCI-2019-08494 | P2 |
Recruiting |
Myelodysplastic Syndrome|Acute Myelomonocytic Leukemia|Chronic Myelomonocytic Leukemia|Acute Monocytic Leukemia|Preleukemia|Juvenile Myelomonocytic Leukemia, |
2026-12-31 |
50% |
2024-10-18 |
Primary Endpoints |
NCT04905316 |
CHORUS | P2 |
Active, not recruiting |
Non-Small-Cell Lung Cancer |
2026-05-01 |
12% |
2025-06-18 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments |
2015-003522-13 |
2015-003522-13 | P2 |
Active, not recruiting |
Familial Mediterranean Fever |
2019-09-09 |
2025-07-04 |
Primary Completion Date|Start Date|Study Completion Date|Treatments |
|
2020-002773-10 |
CANAL | P3 |
Active, not recruiting |
Lung Cancer |
2034-03-14 |
|||
NCT06497491 |
CAN-AOSD-III | P3 |
Completed |
Still's Disease, Adult-Onset |
2025-05-29 |
48% |
2025-08-19 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
NCT04229004 |
PanCAN_Precision Promise | P3 |
Completed |
Pancreatic Cancer|Adenocarcinoma |
2025-02-05 |
2025-04-08 |
||
2015-003491-69 |
2015-003491-69 | P3 |
Active, not recruiting |
Cold Urticaria|Cryopyrin-Associated Periodic Syndromes |
2022-06-16 |
2025-06-28 |
Primary Completion Date|Start Date|Study Completion Date|Treatments|Trial Status |
|
2020-001854-23 |
AMMURAVID | P3 |
Active, not recruiting |
COVID-19 |
2020-08-27 |
2022-03-13 |
Treatments |
|
jRCT2011200022 |
jRCT2011200022 | P3 |
Active, not recruiting |
Still's Disease, Adult-Onset |
2024-05-31 |
|||
NCT04798339 |
MCC-20552 | P2 |
Active, not recruiting |
Preleukemia|Myelodysplastic Syndrome |
2025-05-26 |
50% |
2025-08-07 |
Primary Completion Date|Primary Endpoints|Treatments |
JapicCTI-205213 |
JapicCTI-205213 | P2 |
Active |
Non-Small-Cell Lung Cancer |
2022-01-31 |
|||
JapicCTI-183994 |
JapicCTI-183994 | P3 |
Active |
Non-Small-Cell Lung Cancer |
2023-12-01 |
|||
2024-511490-29-00 |
CACZ885U2301 | P3 |
Active, not recruiting |
Non-Small-Cell Lung Cancer |
2027-06-07 |
60% |
2025-05-02 |
Treatments |
NCT04717635 |
AOSD | P3 |
Completed |
Still's Disease, Adult-Onset |
2025-04-16 |
29% |
2025-05-07 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
01/21/2026 |
News Article |
Variant Bio Appoints Craig T. Basson, MD, PhD, to Advance Genomics-Driven Clinical Programs |
|
12/02/2025 |
News Article |
Ventyx Provides Clinical and Corporate Updates |
|
10/22/2025 |
News Article |
Ventyx Biosciences Announces Positive Topline Results from Phase 2 Study of VTX3232 in Participants with Obesity and Cardiovascular Risk Factors |
|
07/17/2025 |
News Article |
Novartis reports strong Q2 with double-digit sales growth and core margin expansion; raises FY 2025 core operating income guidance |


