Product Description
An orally bioavailable inhibitor of the serine/threonine enzyme hematopoietic progenitor kinase 1 (Hpk1; Hpk-1; mitogen-activated protein kinase kinase kinase kinase 1; MAP4K1; MEKKK1), with potential immunostimulating and antineoplastic activities. Upon oral administration, Hpk1 inhibitor BGB-26808 targets, binds to and inhibits the activity of Hpk1. This inhibits Hpk1-mediated signaling pathways and prevents Hpk1-mediated immunosuppression by preventing the inhibition of T-cell receptors (TCR) signaling and effector T cells, disrupting abnormal cytokine expression, and abrogating the immunosuppressive tumor microenvironment (TME). This may activate a cytotoxic T-lymphocyte (CTL)-mediated immune response against tumor cells. Hpk1, expressed in hematopoietic cells, is a negative regulator of TCR signaling and T- and B-cell activation. (Sourced from: https://www.cancer.gov/publications/dictionaries/cancer-drug/def/hpk1-inhibitor-bgb-26808)
Mechanisms of Action: HPK1 Inhibitor
Novel Mechanism: Yes
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: BeiGene
Company Location:
Company Founding Year: 2010
Additional Commercial Interests: None
Clinical Description
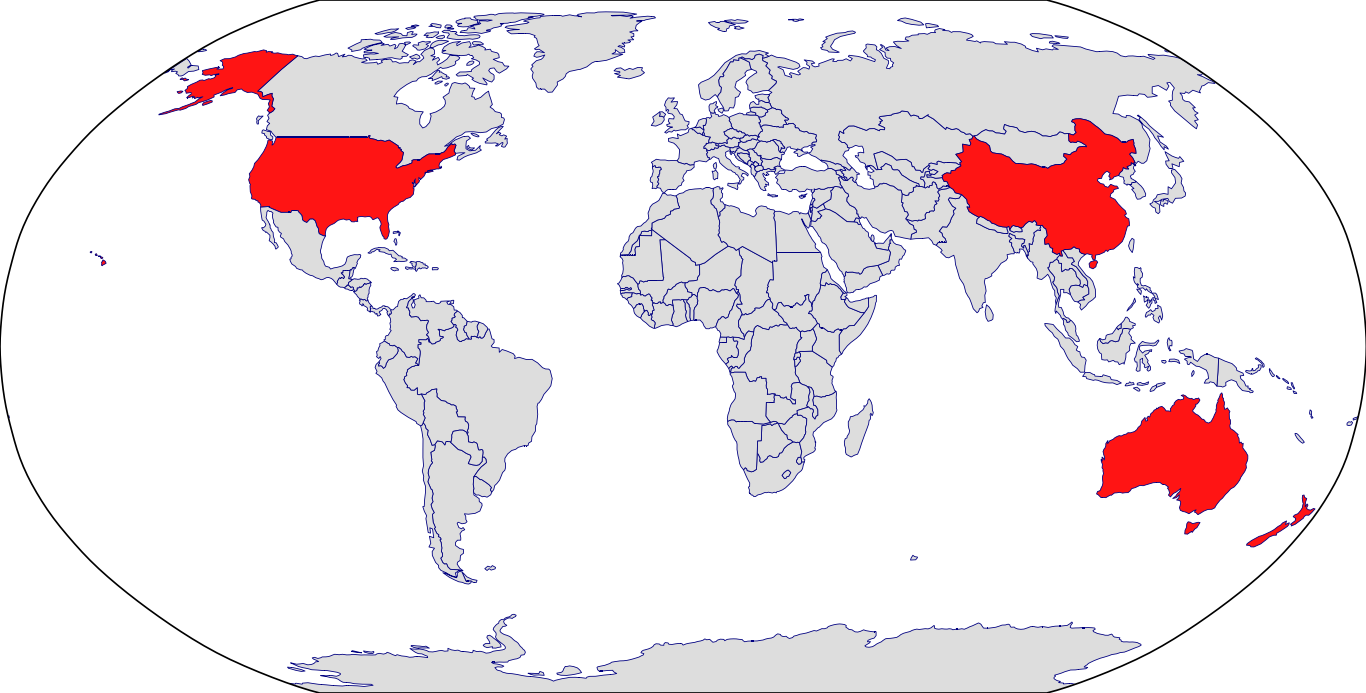
Countries in Clinic: Australia, China, New Zealand, United States
Active Clinical Trial Count: 2
Recent & Upcoming Milestones
Highest Development Phases
Phase 1: Oncology Solid Tumor Unspecified
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05981703 |
BGB-A317-26808-101 | P1 |
Recruiting |
Oncology Solid Tumor Unspecified |
2027-09-30 |
50% |
2025-07-01 |
|
CTR20240210 |
CTR20240210 | P1 |
Recruiting |
Oncology Solid Tumor Unspecified |
None |
50% |
2025-07-27 |
Patient Enrollment|Treatments |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
10/20/2025 |
News Article |
BeOne Medicines Presents New Data on TEVIMBRA in Lung Cancer at ESMO 2025 |


