Product Description
a vasopressin 1a receptor antagonist (Sourced from: https://pubmed.ncbi.nlm.nih.gov/35151410/)
Mechanisms of Action: V1 Antagonist
Novel Mechanism: Yes
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Hoffmann-La Roche
Company Location:
Company Founding Year: 1896
Additional Commercial Interests: None
Clinical Description
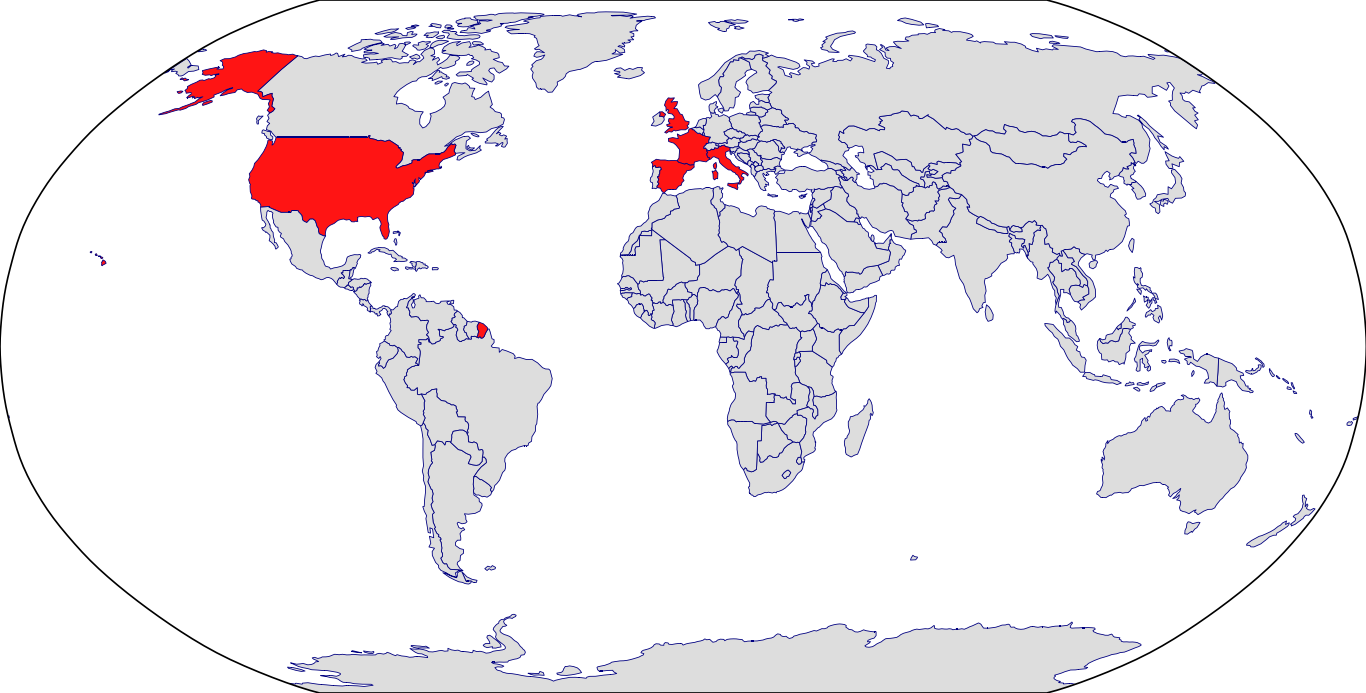
Countries in Clinic: France, Italy, Spain, United Kingdom, United States
Active Clinical Trial Count: 3
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Ischemic Stroke|Stress Disorders, Post-Traumatic
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05310630 |
WC42759 | P2 |
Not yet recruiting |
Ischemic Stroke |
2024-06-27 |
42% |
2022-04-06 |
Primary Endpoints|Treatments |
NCT05401565 |
BN43546 | P2 |
Completed |
Stress Disorders, Post-Traumatic |
2023-10-05 |
29% |
2024-04-19 |
Patient Enrollment|Primary Endpoints|Treatments |
2021-002076-39 |
2021-002076-39 | P2 |
Active, not recruiting |
Ischemic Stroke |
2024-06-10 |
2022-03-13 |
Treatments |


