Product Description
AZD1446 selectively activates the _4_2 subtype of nicotinic acetylcholine receptor. (Sourced from: https://www.alzforum.org/therapeutics/azd1446)
Mechanisms of Action: a4b2 Agonist
Novel Mechanism: Yes
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: AstraZeneca
Company Location: Europe
Company Founding Year: 1999
Additional Commercial Interests: None
Clinical Description
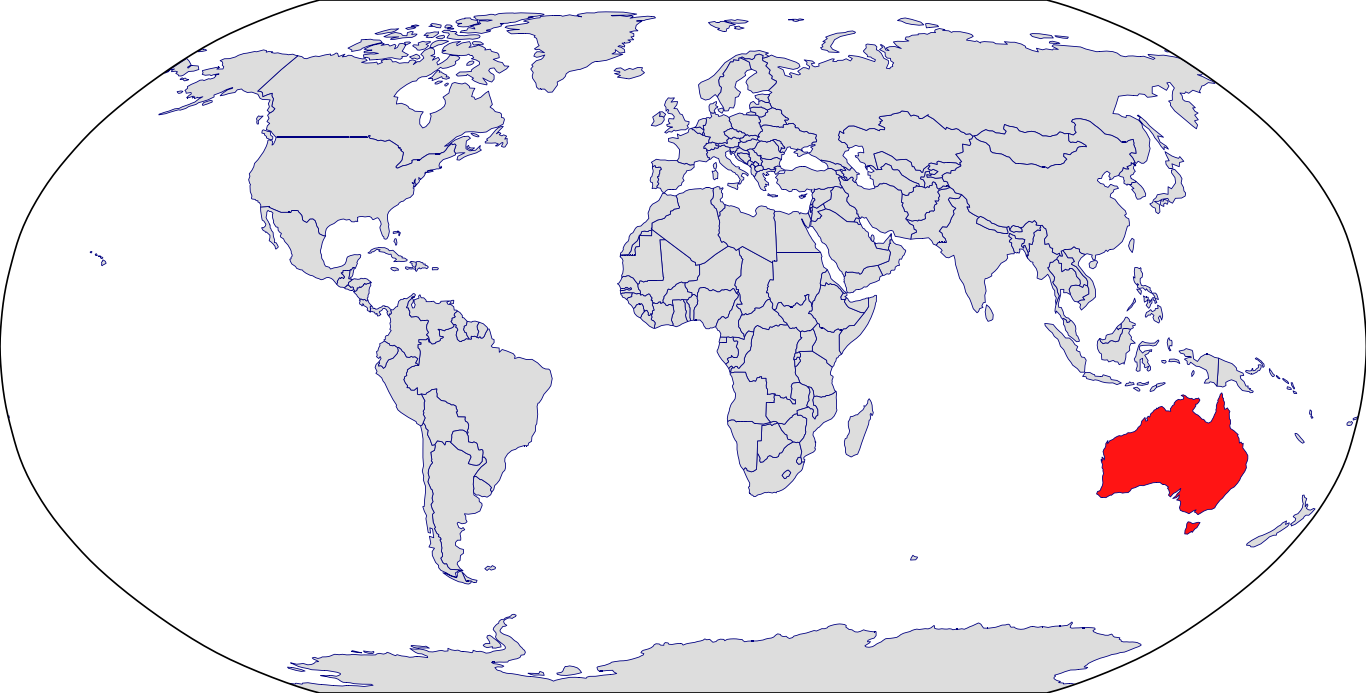
Countries in Clinic: Australia
Active Clinical Trial Count: 1
Recent & Upcoming Milestones
Highest Development Phases
Phase 1: Spinal Cord Injuries
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
ACTRN12621000303842p |
2006-7041-83/hah | P1 |
Not yet recruiting |
Spinal Cord Injuries |
None |
Recent News Events
Date |
Type |
Title |
|---|


