Product Description
Avacincaptad pegol, a polyethylene glycol-conjugated oligonucleotide, is a potent C5 inhibitor delivered via a 100 µl intravitreal injection. (Sourced from: https://iovs.arvojournals.org/article.aspx?articleid=2772902)
Mechanisms of Action: C5 Inhibitor
Novel Mechanism: No
Modality: Nucleic Acid
Route of Administration: Injection
FDA Designation: Breakthrough Therapy - Geographic AtrophyFast Track - Geographic Atrophy|Macular DegenerationPriority Review - Geographic Atrophy *
Approval Status: Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: IVERIC bio
Company Location:
Company Founding Year: 2007
Additional Commercial Interests: DelSiTech
Clinical Description
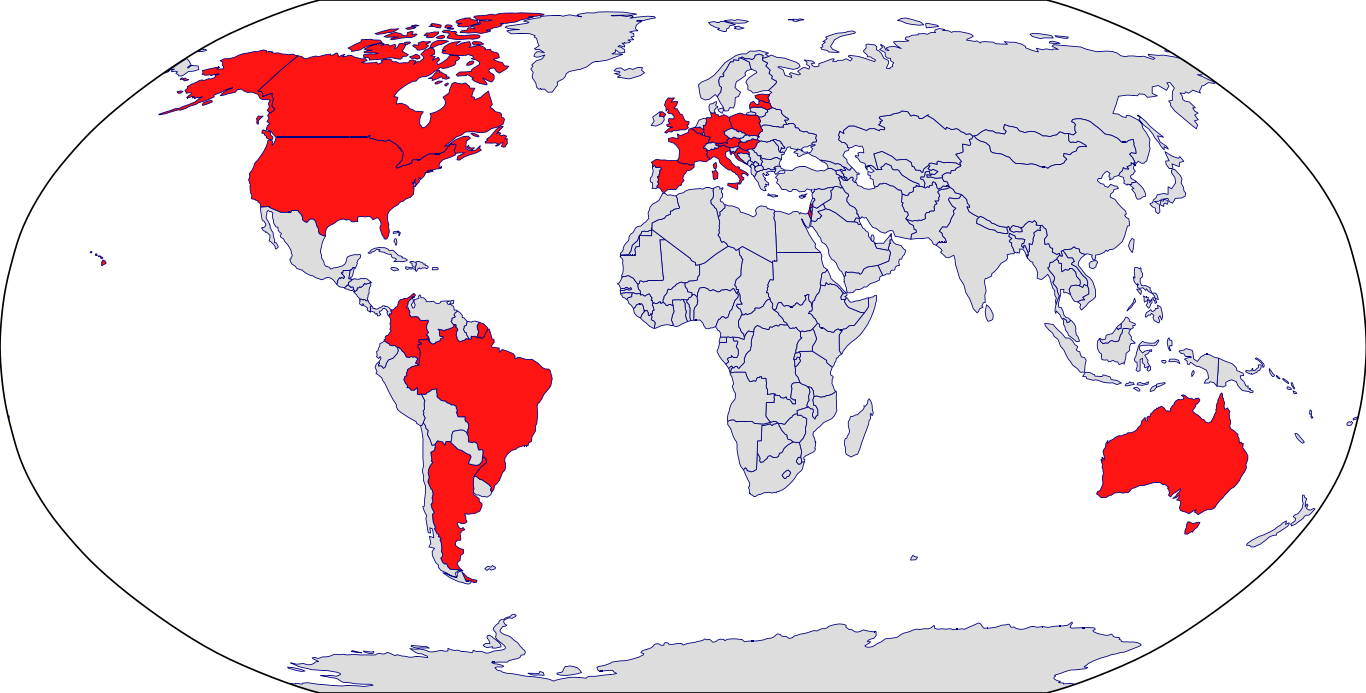
Countries in Clinic: Argentina, Australia, Austria, Belgium, Brazil, Canada, Colombia, Croatia, Czech Republic, France, Germany, Hungary, Israel, Italy, Latvia, Spain, United Kingdom, United States
Active Clinical Trial Count: 4
Recent & Upcoming Milestones
- PDUFA Summary: FDA accepted resubmitted SNDA for Izervay with a target action date on February 26, 2025, for GA treatment.
- FDA accepts ACP NDA filing with priority review for geographic atrophy treatment, with a PDUFA goal date of August 19, 2023.
Highest Development Phases
Phase 3: Geographic Atrophy|Macular Degeneration
Phase 2: Stargardt Disease
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT03364153 |
STGD1 | P2 |
Completed |
Stargardt Disease|Macular Degeneration |
2025-03-31 |
50% |
2025-05-23 |
Primary Endpoints |
NCT05536297 |
GATHER2 | P3 |
Completed |
Macular Degeneration|Geographic Atrophy |
2025-04-10 |
23% |
2025-05-09 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
2017-004783-35 |
2017-004783-35 | P2 |
Active, not recruiting |
Stargardt Disease |
2021-01-10 |
50% |
2022-03-13 |
Treatments |
2024-515185-13-00 |
ISEE2009 | P3 |
Completed |
Geographic Atrophy |
2025-04-10 |
2025-05-02 |
Treatments |


