Product Description
Ataluren is an orally delivered, investigational drug that has the potential to overcome the effects of the nonsense mutation. For Patients With Nonsense Mutation Dystrophinopathy (Sourced from: https://clinicaltrials.gov/ct2/show/NCT01826487)
Mechanisms of Action: Unknown
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Not Approved
Approved Countries: Austria | Belgium | Brazil | Chile | Croatia | Czech | Denmark | Estonia | European Medicines Agency | Finland | France | Germany | Hungary | Iceland | Ireland | Israel | Italy | Korea | Latvia | Lithuania | Netherlands | Norway | Peru | Poland | Portugal | Romania | Russia | Saudi Arabia | Slovakia | Slovenia | Spain | Sweden | Ukraine | United Kingdom
Approved Indications: None
Known Adverse Events: None
Company: PTC Therapeutics
Company Location: Eastern America
Company Founding Year: 1998
Additional Commercial Interests: None
Clinical Description
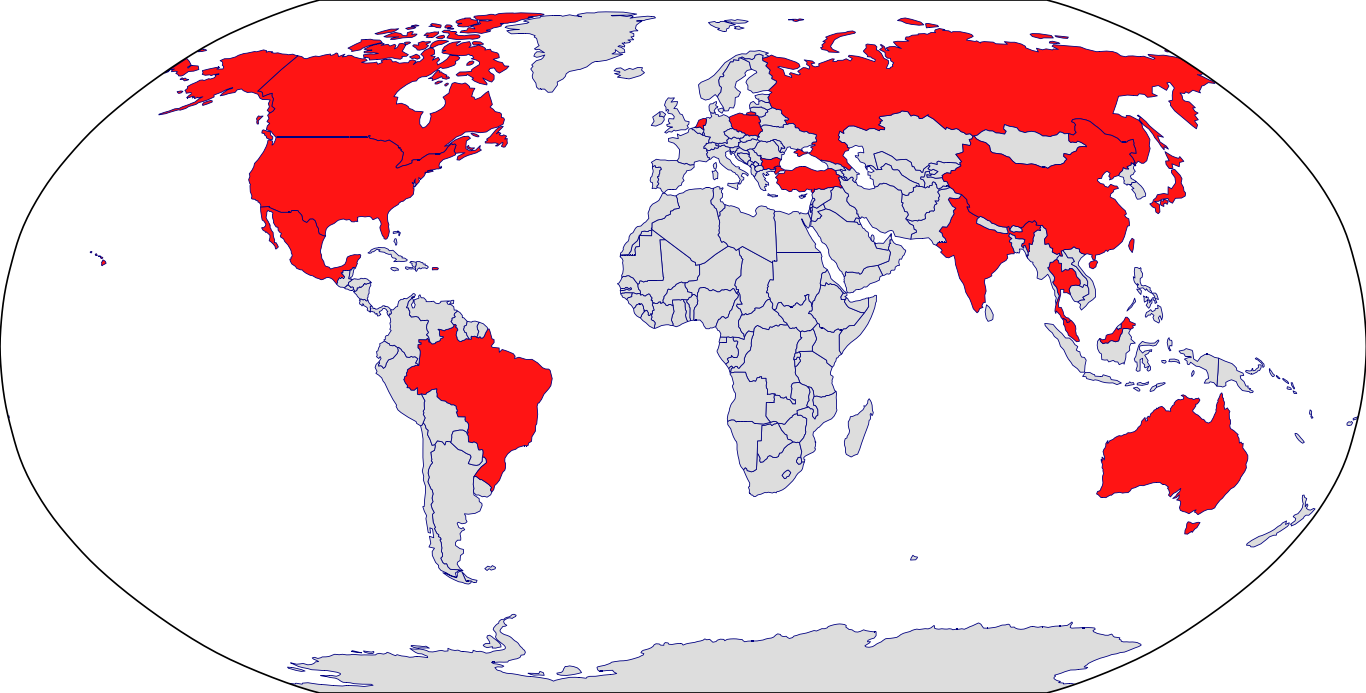
Countries in Clinic: Canada, China, Japan, Netherlands, United States
Active Clinical Trial Count: 7
Recent & Upcoming Milestones
Highest Development Phases
Phase 3: Muscular Dystrophy, Duchenne
Phase 2: Colorectal Cancer|Endometrial Cancer
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT01247207 |
nmDBMD | P3 |
Completed |
Muscular Dystrophy, Duchenne |
2026-02-10 |
50% |
2026-03-10 |
Primary Completion Date|Primary Endpoints |
NCT04014530 |
ATAPEMBRO | P2 |
Recruiting |
Endometrial Cancer|Colorectal Cancer |
2023-06-01 |
10% |
2026-01-09 |
|
2016-001764-11 |
2016-001764-11 | P2 |
Active, not recruiting |
Muscular Dystrophy, Duchenne |
2020-06-05 |
2025-05-06 |
Primary Completion Date|Start Date|Study Completion Date|Treatments |
|
JapicCTI-194632 |
JapicCTI-194632 | P3 |
Active |
Muscular Dystrophy, Duchenne |
2021-09-30 |
|||
jRCT2041200087 |
jRCT2041200087 | P3 |
Recruiting |
Muscular Dystrophy, Duchenne |
2024-01-31 |
|||
CTR20180881 |
CTR20180881 | P3 |
Completed |
Muscular Dystrophy, Duchenne |
2023-07-30 |
2025-04-29 |
Patient Enrollment|Primary Completion Date|Start Date|Study Completion Date|Treatments|Trial Status |
|
NCT04336826 |
nmDMD | P2 |
Completed |
Muscular Dystrophy, Duchenne |
2023-08-07 |
50% |
2023-09-22 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |


