Product Description
ACI-35 is a liposome-based vaccine. The rationale behind it is that the vaccine will treat tauopathy in Alzheimer's disease by eliciting an immune response targeted to certain pathological conformers of phosphorylated tau without also mounting autoimmune B cell or T cell responses against physiological forms of this ubiquitous intracellular protein. The vaccine contains 16 copies of a synthetic tau fragment phosphorylated at the protein's pathological phosphorylation residues S396 and S404 and is anchored into a lipid bilayer. It uses the adjuvant MPLA. (Sourced from: https://www.alzforum.org/therapeutics/aci-35)
Mechanisms of Action: Vaccine, Tau
Novel Mechanism: Yes
Modality: Vaccine
Route of Administration: Injection
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Johnson & Johnson
Company Location: Eastern America
Company Founding Year: 1886
Additional Commercial Interests: None
Clinical Description
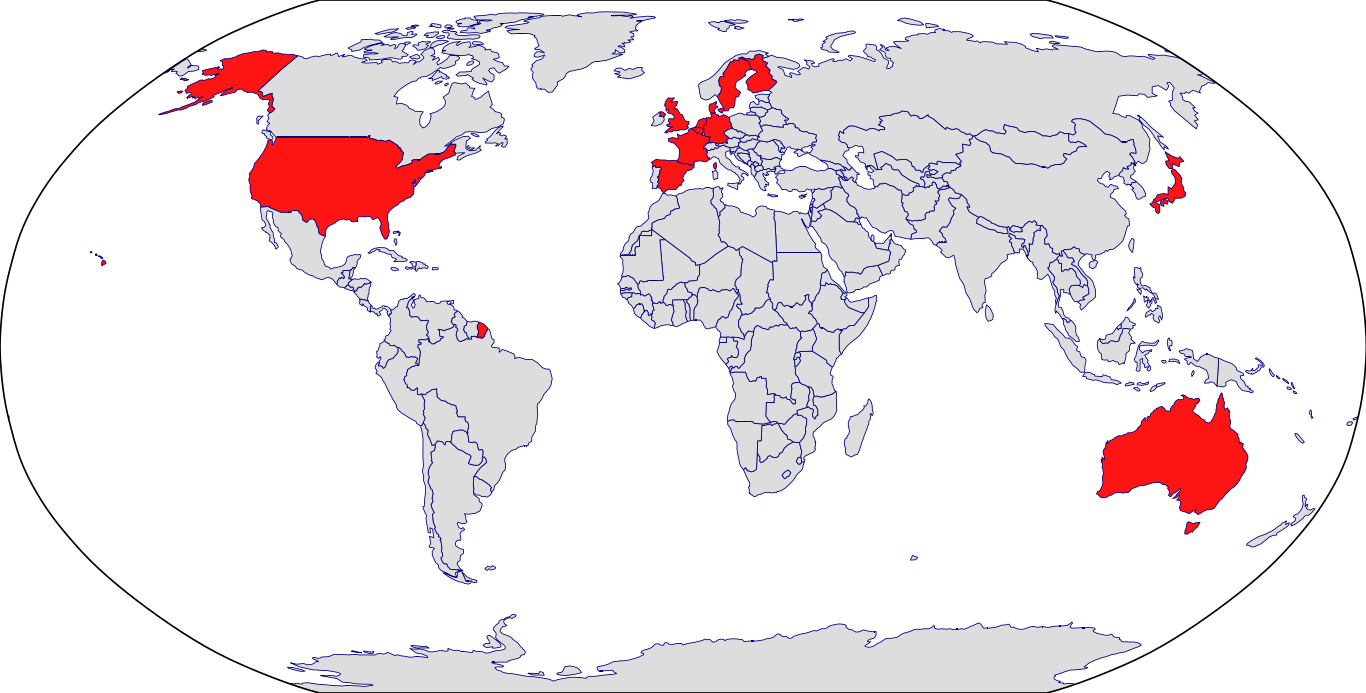
Countries in Clinic: Australia, Belgium, Denmark, Finland, France, Germany, Japan, Netherlands, Spain, Sweden, United Kingdom, United States, Unknown Location
Active Clinical Trial Count: 4
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Alzheimer Disease|Brain Diseases|Cognitive Dysfunction|Tauopathies
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT06544616 |
ReÏain | P2 |
Active, not recruiting |
Alzheimer Disease |
2031-07-17 |
12% |
2026-02-18 |
Primary Endpoints|Study Completion Date|Treatments|Trial Status |
NCT04445831 |
ACI-35-1802 | P2 |
Completed |
Brain Diseases|Alzheimer Disease|Tauopathies|Cognitive Dysfunction |
2023-09-05 |
18% |
2023-10-07 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
2023-505096-68-00 |
64042056ALZ2001 | P2 |
Recruiting |
Alzheimer Disease |
2031-04-30 |
12% |
2025-05-02 |
Treatments |
jRCT2031240382 |
jRCT2031240382 | P2 |
Recruiting |
Alzheimer Disease |
2024-05-31 |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
09/25/2025 |
News Article |
Peer-reviewed results from Phase 1b/2a Trial of Anti-pTau Active Immunotherapy from AC Immune Published in eBioMedicine |
|
03/25/2025 |
News Article |
AC Immune Announces Upcoming Presentations and Industry Symposium on Active Immunotherapies at AD/PD™ 2025 |
|
11/30/2022 |
News Article |
AC Immune's Alzheimer's Disease Vaccine-candidate ACI-35.030 Selected for Further Development |
|
11/30/2022 |
News Article |
AC Immune's Alzheimer's Disease Vaccine-candidate ACI-35.030 Selected for Further Development |


