Product Description
Drospirenone is one of several different progestins that are used in birth control pills. Most birth control pills (combination oral contraceptives) combine a synthetic version of the female hormone progesterone (referred to as a progestin) with a synthetic version of the female hormone estrogen. (Sourced from: https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/information-about-drospirenone)
Mechanisms of Action: MCRA Antagonist
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Approved
Approved Countries: Algeria | Argentina | Australia | Austria | Bangladesh | Belgium | Bosnia | Brazil | Bulgaria | Canada | Chile | Colombia | Croatia | Cyprus | Czech | Denmark | Dominican Republic | Ecuador | Egypt | Estonia | European Medicines Agency | Finland | France | Germany | Greece | Hong Kong | Hungary | Iceland | India | Indonesia | Ireland | Israel | Italy | Japan | Jordan | Korea | Latvia | Lebanon | Lithuania | Luxembourg | Malaysia | Malta | Mexico | Morocco | Netherlands | New Zealand | Norway | Pakistan | Peru | Philippines | Poland | Portugal | Romania | Russia | Saudi Arabia | Serbia | Singapore | Slovakia | Slovenia | South Africa | Spain | Sri Lanka | Sweden | Switzerland | Taiwan | Thailand | Tunisia | Turkey | Ukraine | United Arab Emirates | United Kingdom | United States | Uruguay | Venezuela | Vietnam
Approved Indications: None
Known Adverse Events: None
Company: Bayer
Company Location:
Company Founding Year: 1863
Additional Commercial Interests: None
Clinical Description
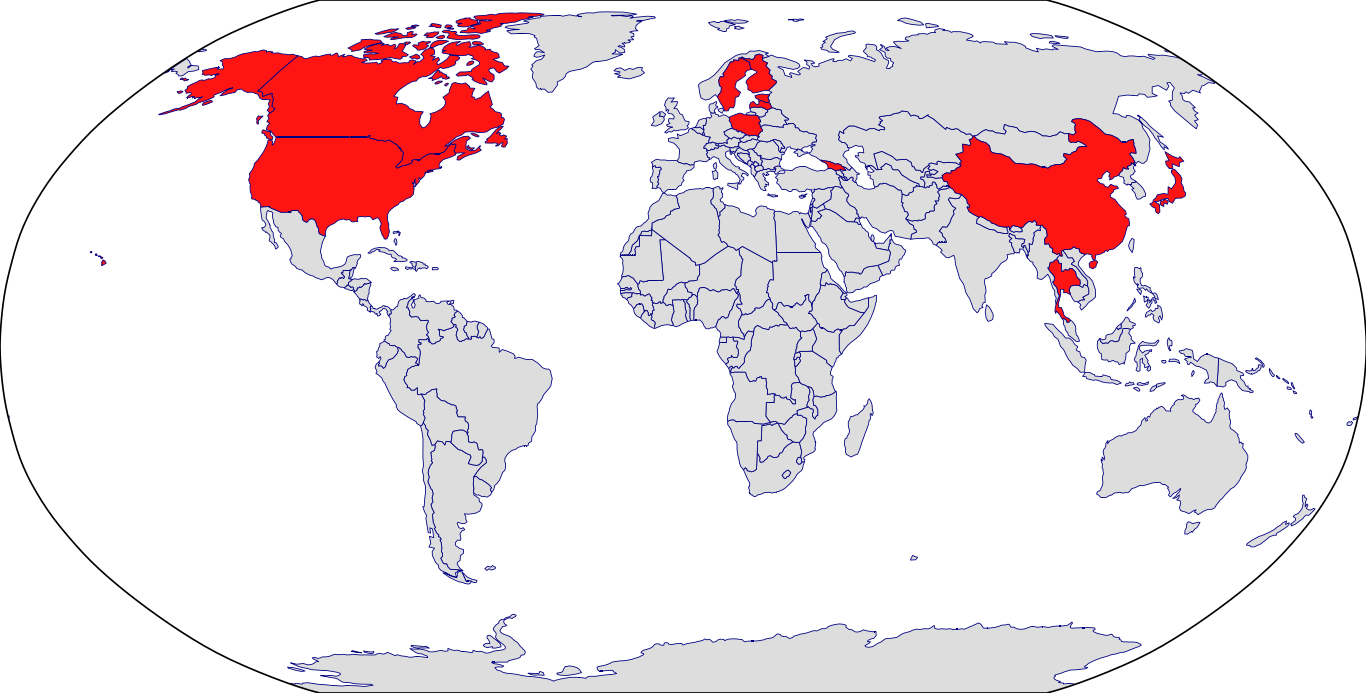
Countries in Clinic: Canada, China, Estonia, Finland, Georgia, Japan, Latvia, Poland, Sweden, Thailand, United States
Active Clinical Trial Count: 9
Recent & Upcoming Milestones
Highest Development Phases
Phase 3: Acute Respiratory Distress Syndrome|Contraception|Endometriosis
Phase 2: Healthy Volunteers
Phase 1: Psoriasis|Pulmonary Fibrosis
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT07181343 |
Yasmin® | P1 |
Completed |
Psoriasis |
2025-06-17 |
2025-09-19 |
Primary Endpoints |
|
NCT05985590 |
IM027-1013 | P1 |
Completed |
Pulmonary Fibrosis |
2023-12-11 |
23% |
2024-01-25 |
Primary Endpoints |
NCT05461573 |
CF113-303 | P3 |
Completed |
Contraception |
2024-08-05 |
58% |
2025-10-09 |
Patient Enrollment|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
NCT04792385 |
MIT-Es001-C303 | P3 |
Completed |
Contraception|Acute Respiratory Distress Syndrome |
2023-11-24 |
15% |
2023-12-23 |
Primary Endpoints |
2021-001424-17 |
2021-001424-17 | P3 |
Completed |
Endometriosis |
2023-09-25 |
2025-05-06 |
Primary Completion Date|Study Completion Date|Treatments|Trial Status |
|
jRCT2031210556 |
jRCT2031210556 | P3 |
Active, not recruiting |
Contraception |
2024-09-30 |
|||
NCT06233071 |
DRE-026-22 | P1 |
Recruiting |
Healthy Volunteers |
2024-04-04 |
12% |
2024-03-13 |
Primary Endpoints|Treatments|Trial Status |
jRCT2071240132 |
jRCT2071240132 | P2 |
Not yet recruiting |
Healthy Volunteers |
2026-01-31 |
|||
2019-003002-27 |
2019-003002-27 | P3 |
Active, not recruiting |
Contraception |
2023-04-14 |
15% |
2022-03-13 |
Treatments |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
03/11/2026 |
News Article |
New Survey of 120,000+ Americans Reveals Massive Demand for Flexible, At-Home Earning — Online Tasks Rank as the #1 Preferred Side Hustle Category |
|
03/10/2026 |
News Article |
Maybelline New York Launches "Record Breakers Go Sky High" Campaign Starring Aisha Bowe, Alex Aster, and Ilona Maher |
|
03/09/2026 |
News Article |
Baked by Melissa Launches First-Ever Beauty Brand Collaboration With essie |
|
02/10/2026 |
News Article |
Daea Wins First Place in Pamela Anderson's Sonsie x Shopify Mindful Beauty Award |


