Product Description
Margetuximab-cmkb injection is used to treat metastatic (cancer that has spread) HER2-positive breast cancer in patients who have received two or more anti-HER2 breast cancer treatments, and at least one treatment for metastatic disease. (Sourced from: https://www.mayoclinic.org/drugs-supplements/margetuximab-cmkb-intravenous-route/description/drg-20506401)
Mechanisms of Action: Unknown
Novel Mechanism: No
Modality: Antibody
Route of Administration: Intravenous
FDA Designation: Orphan Drug - Gastrointestinal Cancer *
Approval Status: Approved
Approved Countries: Israel | United States
Approved Indications: None
Known Adverse Events: None
Company: MacroGenics
Company Location: Eastern America
Company Founding Year: 2000
Additional Commercial Interests: None
Clinical Description
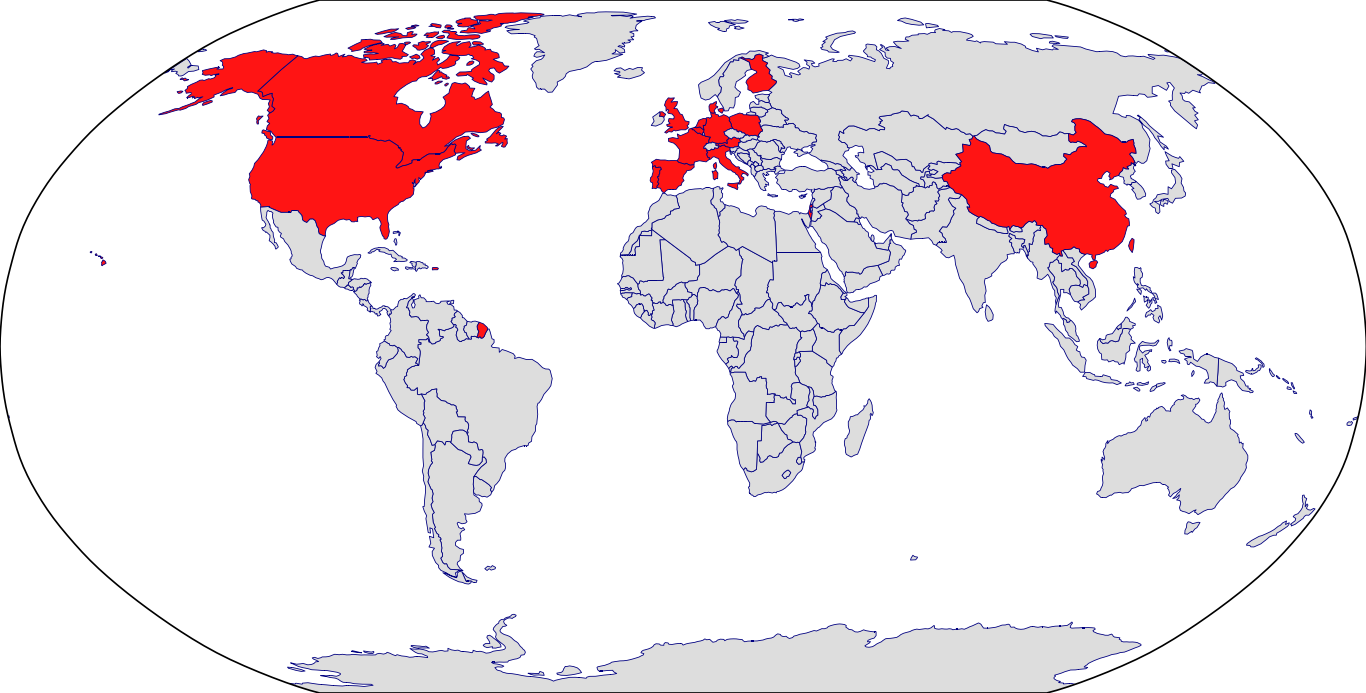
Countries in Clinic: China, Germany, Italy, Korea, Poland, Singapore, Taiwan, United Kingdom, United States
Active Clinical Trial Count: 3
Recent & Upcoming Milestones
- PDUFA target action date for Margetuximab BLA is December 18, 2020, in treating metastatic HER2-positive breast cancer.
Highest Development Phases
Phase 3: Esophageal Cancer|Gastrointestinal Cancer
Phase 2: Breast Cancer
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT04425018 |
MARGOT | P2 |
Active, not recruiting |
Breast Cancer |
2024-10-24 |
12% |
2025-06-27 |
Primary Endpoints |
NCT04082364 |
MAHOGANY | P3 |
Completed |
Esophageal Cancer|Gastrointestinal Cancer |
2024-01-15 |
19% |
2025-04-23 |
|
CTR20200222 |
CTR20200222 | P2 |
Active, not recruiting |
Breast Cancer |
None |
2025-04-29 |
Patient Enrollment|Start Date|Treatments|Trial Status |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
10/22/2024 |
News Article |
MacroGenics Enters Into Agreement With TerSera Therapeutics for the Sale of MARGENZA® |
|
07/30/2024 |
News Article |
MacroGenics Announces Achievement of $100 Million in Milestones Related to Retifanlimab Collaboration with Incyte |
|
07/30/2024 |
News Article |
MacroGenics Provides Vobramitamab Duocarmazine Update |
|
04/03/2024 |
News Article |
MacroGenics Provides Phase 2 TAMARACK Study Early Interim Safety Data and Plans for Future Disclosures |


