Product Description
An enzyme that breaks down a substance in the body called hyaluronic acid. Hyaluronic acid is found throughout the body in connective tissue, skin, and fluids in the joints and inside the eye. It causes fluids to have a jelly-like thickness, which can help moisten and protect tissues and joints. In medicine, hyaluronidase is made in the laboratory and given with other drugs by injection under the skin to treat certain conditions. Using hyaluronidase to break down hyaluronic acid helps make the fluids and connective tissue thinner so the drugs can move more easily into the tissues. (Sourced from: https://www.cancer.gov/publications/dictionaries/cancer-terms/def/hyaluronidase)
Mechanisms of Action: Glycosaminoglycan Hydrolyzer
Novel Mechanism: No
Modality: Peptide/Protein
Route of Administration: Subcutaneous
FDA Designation: *
Approval Status: Approved
Approved Countries: Argentina | Australia | Austria | Bangladesh | Belgium | Brazil | Canada | Chile | China | Colombia | Croatia | Czech | Denmark | Dominican Republic | Egypt | Estonia | European Medicines Agency | Finland | France | Germany | Greece | Hong Kong | Hungary | Iceland | India | Ireland | Israel | Italy | Korea | Latvia | Lithuania | Malta | Mexico | Netherlands | New Zealand | Norway | Pakistan | Peru | Philippines | Poland | Portugal | Romania | Russia | Saudi Arabia | Singapore | Slovakia | Slovenia | Spain | Sweden | Switzerland | Taiwan | Turkey | Ukraine | United Arab Emirates | United Kingdom | United States | Venezuela | Vietnam
Approved Indications: None
Known Adverse Events: None
Company: Bausch & Lomb
Company Location: Eastern America
Company Founding Year: 1853
Additional Commercial Interests: None
Clinical Description
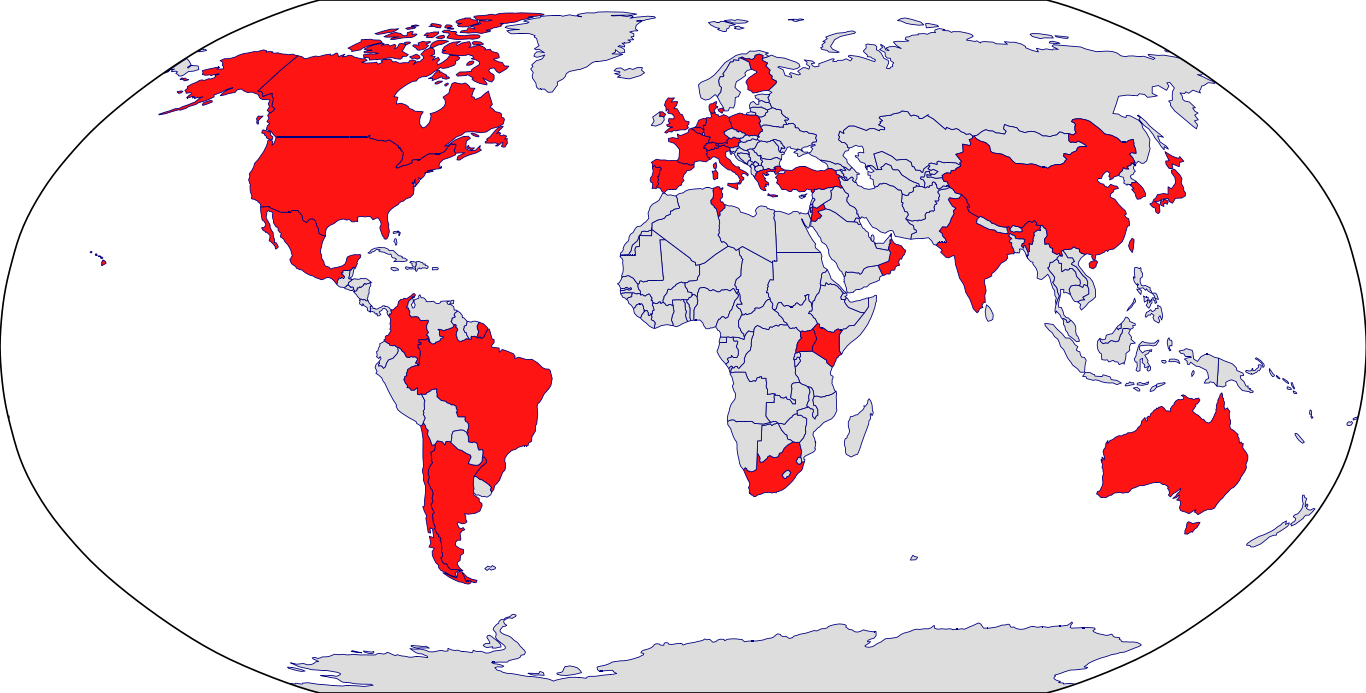
Countries in Clinic: Argentina, Australia, Austria, Belgium, Brazil, Canada, Chile, China, Colombia, Czech Republic, Denmark, Finland, France, Germany, Greece, Hong Kong, India, Italy, Japan, Jordan, Kenya, Korea, Mexico, Netherlands, Norway, Oman, Poland, Portugal, Singapore, South Africa, South Korea, Spain, Switzerland, Taiwan, Tunisia, Turkey, Uganda, United Kingdom, United States, Unknown Location
Active Clinical Trial Count: 21
Recent & Upcoming Milestones
- Clinical Outcomes Reported - Takeda presented P3 Chronic Inflammatory Demyelinating Polyradiculoneuropathy results on 2024-06-30 for Hyaluronidase
Highest Development Phases
Phase 3: Back Pain|Breast Cancer|Chronic Inflammatory Demyelinating Polyradiculoneuropathy|Deficiency Diseases|Edema
Phase 2: Graves Disease|Graves Ophthalmopathy|Nose Cancer|Oncology Solid Tumor Unspecified|Thyroid Eye Disease
Phase 1: Acute Lymphoid Leukemia|Acute Myeloid Leukemia|Anemia, Aplastic|B-Cell Marginal Zone Lymphoma|Chronic Lymphoid Leukemia|Chronic Myeloid Leukemia|Follicular Lymphoma|Healthy Volunteers|Lymphoma, Non-Hodgkin|Multiple Myeloma|Myelodysplastic Syndrome|Myelofibrosis|Myelogenous, Chronic, BCR-ABL Positive Leukemia|Precursor Cell Lymphoblastic Leukemia-Lymphoma|Preleukemia|Stem Cell Transplant
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT06068985 |
CHERRY-PICK | P2 |
Active, not recruiting |
Breast Cancer |
2026-04-01 |
12% |
2026-02-13 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
2024-517450-95-00 |
TAK-881-3003 | P3 |
Not yet recruiting |
Chronic Inflammatory Demyelinating Polyradiculoneuropathy |
2030-05-30 |
24% |
||
2022-003501-29 |
2022-003501-29 | P3 |
Active, not recruiting |
Deficiency Diseases |
2029-01-24 |
2025-07-05 |
Primary Completion Date|Start Date|Study Completion Date|Treatments|Trial Status |
|
NCT06604546 |
SHBJ-KJ017-001 | P3 |
Completed |
Edema|Back Pain |
2024-05-05 |
2024-09-20 |
Primary Endpoints|Treatments |
|
NCT04311606 |
AcTED | P2 |
Completed |
Graves Ophthalmopathy|Graves Disease|Thyroid Eye Disease |
2024-12-17 |
12% |
2026-01-01 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
NCT04632992 |
MyTACTIC | P2 |
Completed |
Oncology Solid Tumor Unspecified |
2023-12-04 |
44% |
2025-01-09 |
Primary Endpoints|Treatments |
NCT04817956 |
IMPRESS-N | P2 |
Recruiting |
Nose Cancer |
2040-03-30 |
2025-08-27 |
Primary Endpoints |
|
NCT04447716 |
19P.835 | P1 |
Active, not recruiting |
Follicular Lymphoma|B-Cell Marginal Zone Lymphoma|Lymphoma, Non-Hodgkin |
2024-05-16 |
12% |
2025-09-16 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments |
NCT06398457 |
J2450 | P1 |
Recruiting |
Chronic Lymphoid Leukemia|Myelodysplastic Syndrome|Precursor Cell Lymphoblastic Leukemia-Lymphoma|Multiple Myeloma|Preleukemia|Acute Myeloid Leukemia|Myelogenous, Chronic, BCR-ABL Positive Leukemia|Stem Cell Transplant|Chronic Myeloid Leukemia|Myelofibrosis|Acute Lymphoid Leukemia|Anemia, Aplastic |
2026-09-01 |
12% |
2024-09-26 |
Primary Endpoints|Start Date |
2022-502046-28-00 |
WO44263 | P3 |
Recruiting |
Breast Cancer |
2028-12-31 |
50% |
2025-05-02 |
Treatments |
NCT05894239 |
WO44263 | P3 |
Recruiting |
Breast Cancer |
2026-09-28 |
50% |
2025-02-15 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments |
NCT05132582 |
HER2CLIMB-05 | P3 |
Active, not recruiting |
Breast Cancer |
2025-09-05 |
25% |
2025-10-08 |
Primary Completion Date|Primary Endpoints|Treatments |
2016-000374-37 |
2016-000374-37 | P3 |
Completed |
Chronic Inflammatory Demyelinating Polyradiculoneuropathy |
2023-07-04 |
31% |
2025-05-06 |
Primary Completion Date |
NCT05139225 |
NCT05139225 | P1 |
Active, not recruiting |
Multiple Myeloma |
2026-10-01 |
12% |
2025-11-05 |
Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments |
NCT05922813 |
BMI2004-SIT-01 | P1 |
Completed |
Healthy Volunteers |
2023-03-10 |
2023-06-30 |
Primary Endpoints|Treatments |
|
CTR20251812 |
CTR20251812 | P1 |
Active, not recruiting |
Healthy Volunteers |
None |
2025-11-16 |
Patient Enrollment|Start Date|Treatments|Trial Status |
|
2019-005019-18 |
2019-005019-18 | P2 |
Active, not recruiting |
Breast Cancer |
2025-07-31 |
|||
NCT06172127 |
DEMETHER | P2 |
Recruiting |
Breast Cancer |
2026-11-01 |
12% |
2024-08-02 |
Primary Endpoints|Start Date|Treatments|Trial Status |
2022-501152-28-00 |
GBG 105 | P2 |
Recruiting |
Breast Cancer |
2026-03-31 |
2025-05-02 |
Treatments |
|
2023-507495-48-00 |
CO42867 | P2 |
Recruiting |
Breast Cancer |
2024-05-23 |
12% |
2025-05-02 |
Treatments |
jRCT2031250577 |
jRCT2031250577 | P3 |
Recruiting |
Breast Cancer |
2032-12-28 |


