Product Description
Concizumab is a monoclonal, humanized IgG4 antibody specific for the Kunitz-2 domain of Tissue Factor Pathway Inhibitor (TFPI). Preclinical studies in vitro or on animal models and in vivo have demonstrated the ability of concizumab to restore thrombin generation, promoting the establishment of a procoagulant action; all these results were subsequently confirmed in the studies of EXPLORER program. (Sourced from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9020573/#:~:text=Concizumab%2C%20an%20anti%2Dtissue%20factor,Hemophilia.)
Mechanisms of Action: SPINT2 Inhibitor
Novel Mechanism: No
Modality: Antibody
Route of Administration: Subcutaneous
FDA Designation: *
Approval Status: Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Novo Nordisk
Company Location: Europe
Company Founding Year: 1923
Additional Commercial Interests: None
Clinical Description
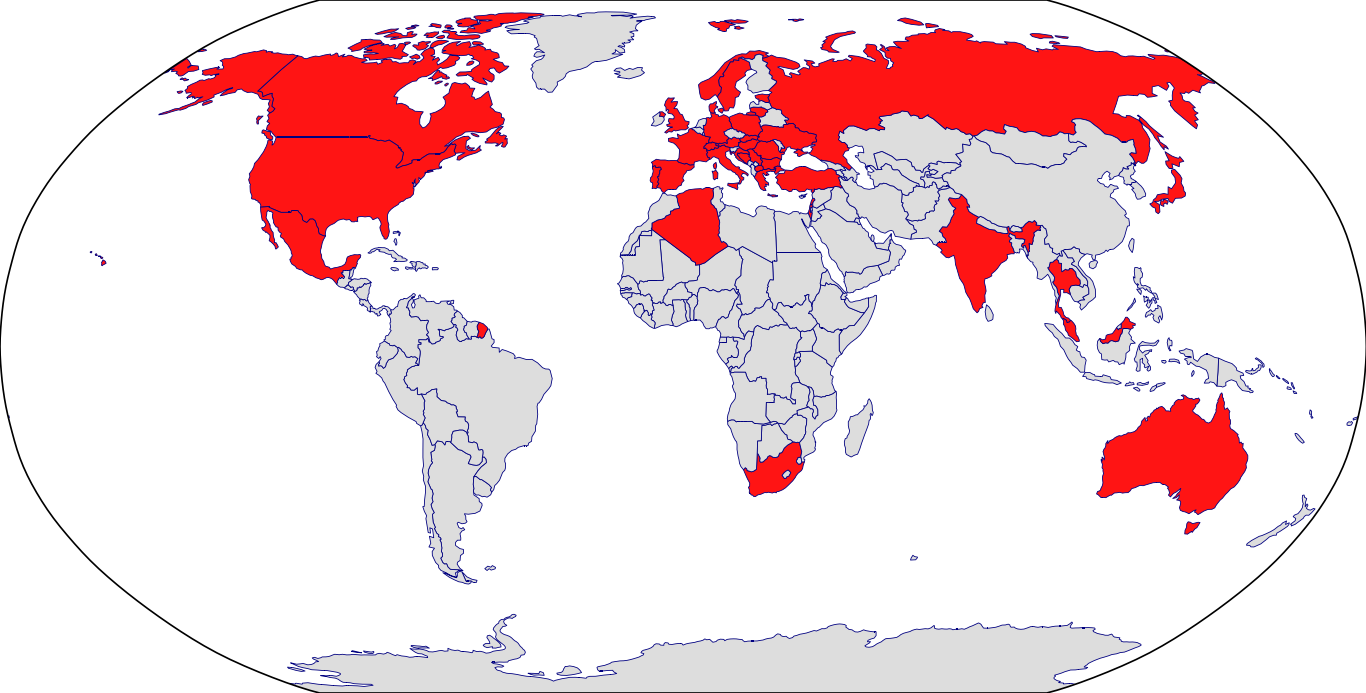
Countries in Clinic: Algeria, Australia, Austria, Bosnia, Bulgaria, Canada, Croatia, Czech Republic, Denmark, Estonia, France, Germany, Greece, Hungary, India, Israel, Italy, Japan, Korea, Lebanon, Lithuania, Malaysia, Mexico, North Macedonia, Norway, Poland, Portugal, Romania, Russia, Serbia, Slovakia, South Africa, Spain, Sweden, Switzerland, Thailand, Turkey, Ukraine, United Kingdom, United States, Unknown Location
Active Clinical Trial Count: 9
Recent & Upcoming Milestones
- Clinical Outcomes Reported - Novo Nordisk presented P3 Hemophilia A|Hemophilia B results on 2024-12-07 for Concizumab
Highest Development Phases
Phase 3: Hemophilia A|Hemophilia B|Injuries/wounds Unspecified
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05135559 |
Explorer10 | P3 |
Active, not recruiting |
Hemophilia A |
2026-04-21 |
37% |
2025-10-11 |
|
2023-506832-33-00 |
NN7415-4311 | P3 |
Active, not recruiting |
Injuries/wounds Unspecified |
2025-12-31 |
2025-05-02 |
Treatments |
|
NCT04082429 |
explorer8 | P3 |
Active, not recruiting |
Hemophilia B|Hemophilia A |
2022-07-12 |
31% |
2025-05-28 |
Primary Endpoints |
2023-506925-13-00 |
NN7415-4616 | P3 |
Recruiting |
Injuries/wounds Unspecified |
2029-11-02 |
2025-05-02 |
Treatments |
|
2023-506831-13-00 |
NN7415-4307 | P3 |
Active, not recruiting |
Injuries/wounds Unspecified |
2026-06-09 |
2025-05-02 |
Treatments |
|
JapicCTI-195046 |
JapicCTI-195046 | P3 |
Active |
Hemophilia A |
2023-03-10 |
|||
jRCT2080224958 |
jRCT2080224958 | P3 |
Completed |
Unknown |
2023-03-10 |
|||
JapicCTI-195045 |
JapicCTI-195045 | P3 |
Active |
Hemophilia A |
2023-03-10 |
|||
NCT04083781 |
explorer7 | P3 |
Active, not recruiting |
Hemophilia B|Hemophilia A |
2021-12-27 |
28% |
2025-04-30 |
Patient Enrollment|Primary Endpoints|Study Completion Date |


