Product Description
BG-T187 is an anti-EGFR×c-MET×c-MET tri-specific antibody that is registered as a monotherapy or in combo with the other therapeutic agents (not disclosed) in the phase 1 trial (NCT06598800). (Sourced from: https://www.cnbio.xyz/2024/09/beigene-has-filed-ind-application-for-BG-T187.html?m=1)
Mechanisms of Action: EGFR Inhibitor, c-MET Inhibitor
Novel Mechanism: No
Modality: Trispecific Antibody
Route of Administration: N/A
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Company: BeOne Medicines
Company Location:
Company Founding Year: None
Additional Commercial Interests: None
Clinical Description
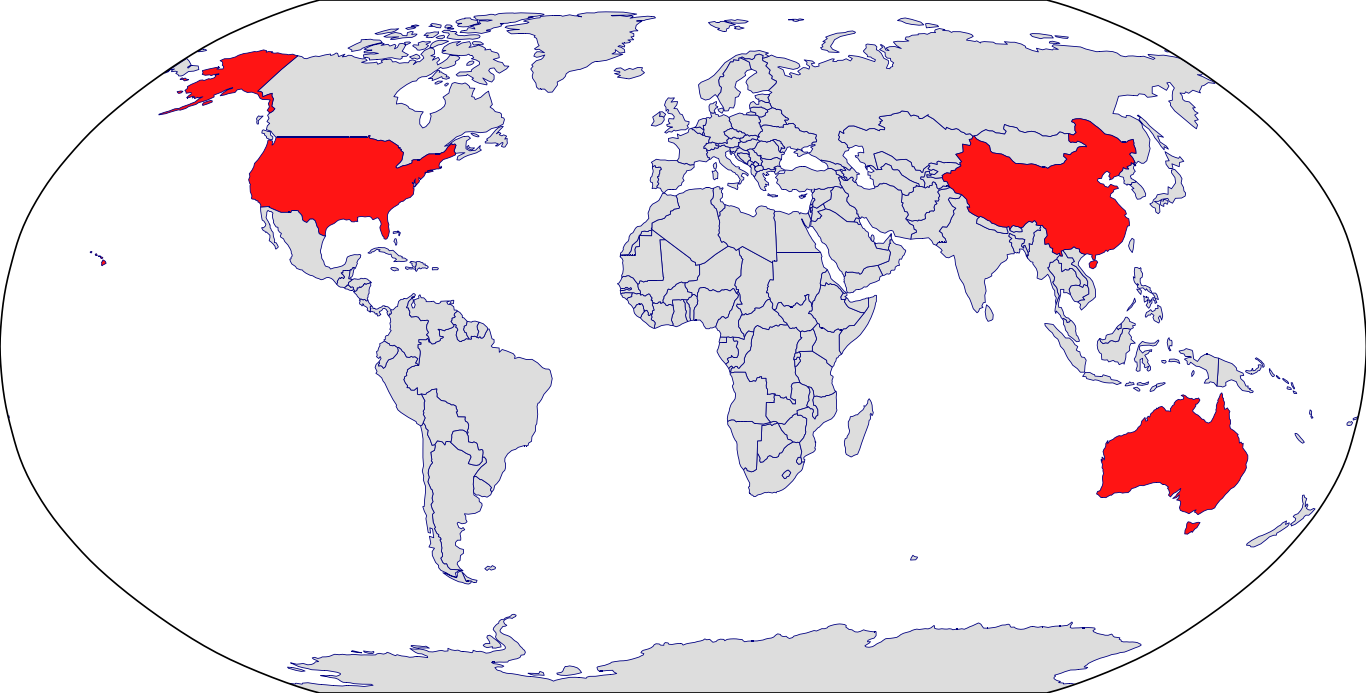
Countries in Clinic: Australia, China, South Korea, United States
Active Clinical Trial Count: 1
Recent & Upcoming Milestones
Highest Development Phases
Phase 1: Oncology Solid Tumor Unspecified
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT06598800 |
BG-T187-101 | P1 |
Recruiting |
Oncology Solid Tumor Unspecified |
2028-09-30 |
50% |
2026-05-23 |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
02/27/2025 |
News Article |
BeiGene Announces Fourth Quarter and Full Year 2024 Financial Results and Business Updates |
|
11/12/2024 |
News Article |
BeiGene Announces Third Quarter 2024 Financial Results and Corporate Updates |


