Product Description
KFA-115, also known as NVP-KFA115, is an immunomodulatory agent being developed by Novartis for the treatment of select advanced cancers. (Sourced from: https://clinicaltrials.gov/ct2/show/NCT05544929?term=KFA-115&draw=2&rank=1)
Mechanisms of Action: Immunomodulator
Novel Mechanism: No
Modality: Unknown
Route of Administration: Oral
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Novartis
Company Location: Europe
Company Founding Year: 1996
Additional Commercial Interests: None
Clinical Description
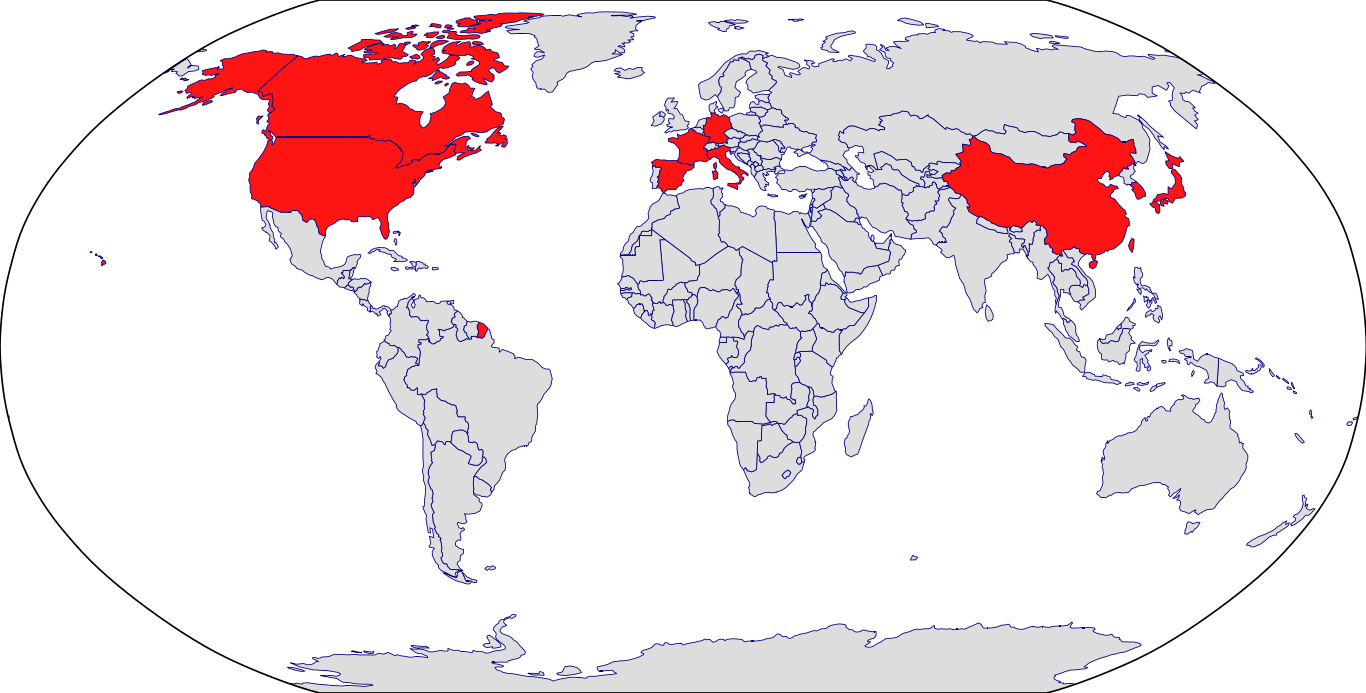
Countries in Clinic: Canada, China, France, Germany, Hong Kong, Italy, Japan, Korea, Singapore, South Korea, Spain, Taiwan, United States, Unknown Location
Active Clinical Trial Count: 3
Recent & Upcoming Milestones
Highest Development Phases
Phase 1: Anus Cancer|Clear Cell Adenocarcinoma|Colorectal Cancer|Cutaneous Squamous Cell Carcinoma|Esophageal Cancer|Gastrointestinal Cancer|Head and Neck Cancer|Kidney Cancer|Melanoma|Mesothelioma|Nasopharyngeal Cancer|Non-Small-Cell Lung Cancer|Oncology Solid Tumor Unspecified|Ovarian Cancer|Renal Cell Carcinoma|Skin Cancer|Squamous Cell Carcinoma|Thymoma|Thymus Cancer|Triple Negative Breast Cancer
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05544929 |
CKFA115A12101 | P1 |
Active, not recruiting |
Cutaneous Squamous Cell Carcinoma|Nasopharyngeal Cancer|Thymoma|Gastrointestinal Cancer|Kidney Cancer|Triple Negative Breast Cancer|Skin Cancer|Ovarian Cancer|Colorectal Cancer|Squamous Cell Carcinoma|Melanoma|Non-Small-Cell Lung Cancer|Head and Neck Cancer|Mesothelioma|Esophageal Cancer|Renal Cell Carcinoma|Thymus Cancer|Clear Cell Adenocarcinoma|Anus Cancer |
2027-09-01 |
12% |
2026-01-07 |
Patient Enrollment|Primary Endpoints|Treatments|Trial Status |
jRCT2031230410 |
jRCT2031230410 | P1 |
Not yet recruiting |
Oncology Solid Tumor Unspecified |
2025-12-31 |
|||
CTR20250770 |
CTR20250770 | P1 |
Active, not recruiting |
Oncology Solid Tumor Unspecified |
None |
2025-12-21 |
Recent News Events
Date |
Type |
Title |
|---|


