Product Description
OTT166 is a novel small molecule selective inhibitor that OcuTerra has purpose engineered to have the required physiochemical characteristics to be able to reach the retina from eye drop application. (Sourced from: https://www.ocuterratx.com/pipeline)
Mechanisms of Action: aVb3 Inhibitor
Novel Mechanism: Yes
Modality: Small Molecule
Route of Administration: Ocular, Topical, Ophthalmic
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: OcuTerra Therapeutics, Inc.
Company Location:
Company Founding Year: None
Additional Commercial Interests: None
Clinical Description
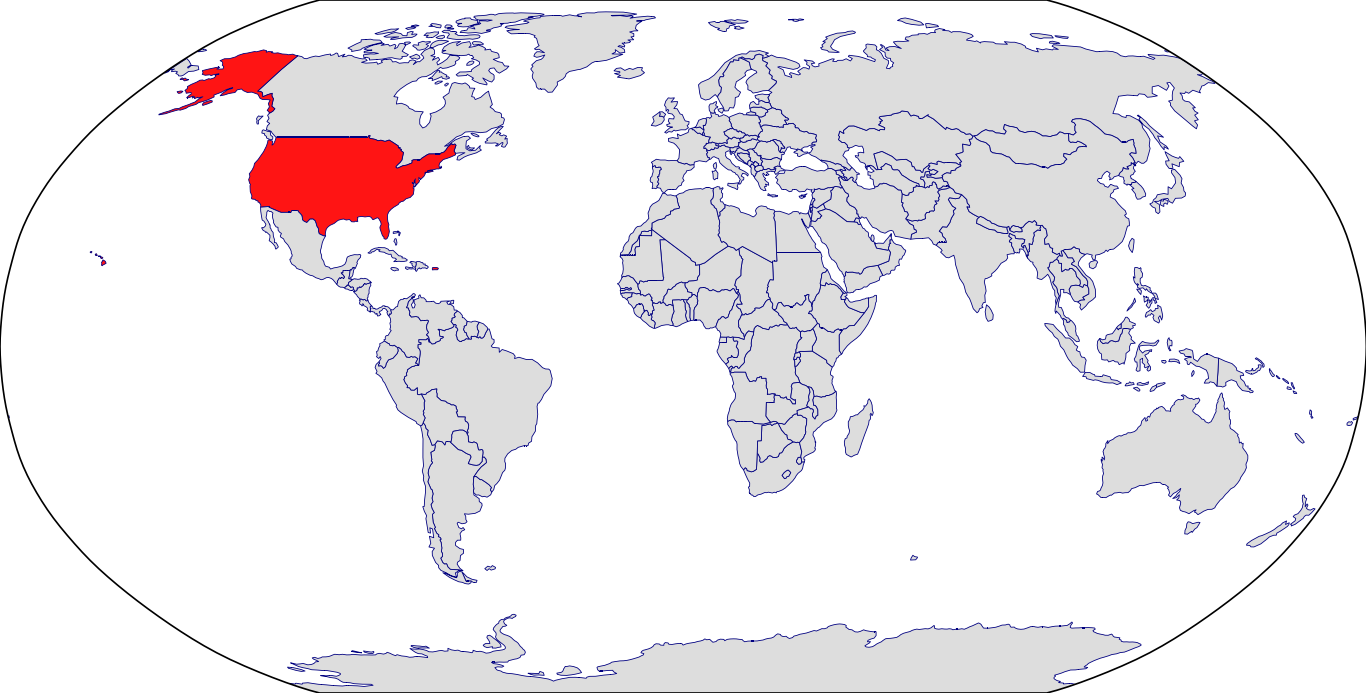
Countries in Clinic: Puerto Rico, United States
Active Clinical Trial Count: 1
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Diabetic Retinopathy
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05409235 |
OTT166-201 | P2 |
Completed |
Diabetic Retinopathy |
2023-12-29 |
50% |
2024-01-11 |
Primary Endpoints |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
01/04/2024 |
News Article |
OcuTerra Therapeutics to Present at 42nd Annual J.P. Morgan Healthcare Conference |
|
09/19/2023 |
News Article |
OcuTerra President and CEO Kerrie Brady Named to 2023 PharmaVoice 100 List of Most Inspiring Leaders |
|
06/01/2023 |
News Article |
OcuTerra Appoints Bill Steinkrauss as Chief Financial Officer |
|
11/22/2022 |
News Article |
OcuTerra to Participate in Upcoming Investor Conferences |
|
10/01/2022 |
PubMed |
The Safety and Biological Activity of OTT166, a Novel Topical Selective Integrin Inhibitor for the Treatment of Diabetic Eye Disease: A Phase 1b Study. |
|
09/01/2020 |
PubMed |
Anti-integrin therapy for retinovascular diseases. |


