Product Description
CYB003 has the potential to effectively treat major depressive disorder (MDD) and alcohol use disorder (AUD). CYB003 is a deuterated psilocybin analog designed to achieve less variability in plasma levels, faster onset of action, shorter duration of effect and potentially better tolerability versus oral psilocybin, for an overall better outcome for patients. (Sourced from: https://cybin.com/cyb003/)
Mechanisms of Action: 5-HT2A Agonist
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: N/A
FDA Designation: Breakthrough Therapy - Depressive Disorder|Depressive Disorder, Major *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Cybin
Company Location: Eastern America
Company Founding Year: 2019
Additional Commercial Interests: None
Clinical Description
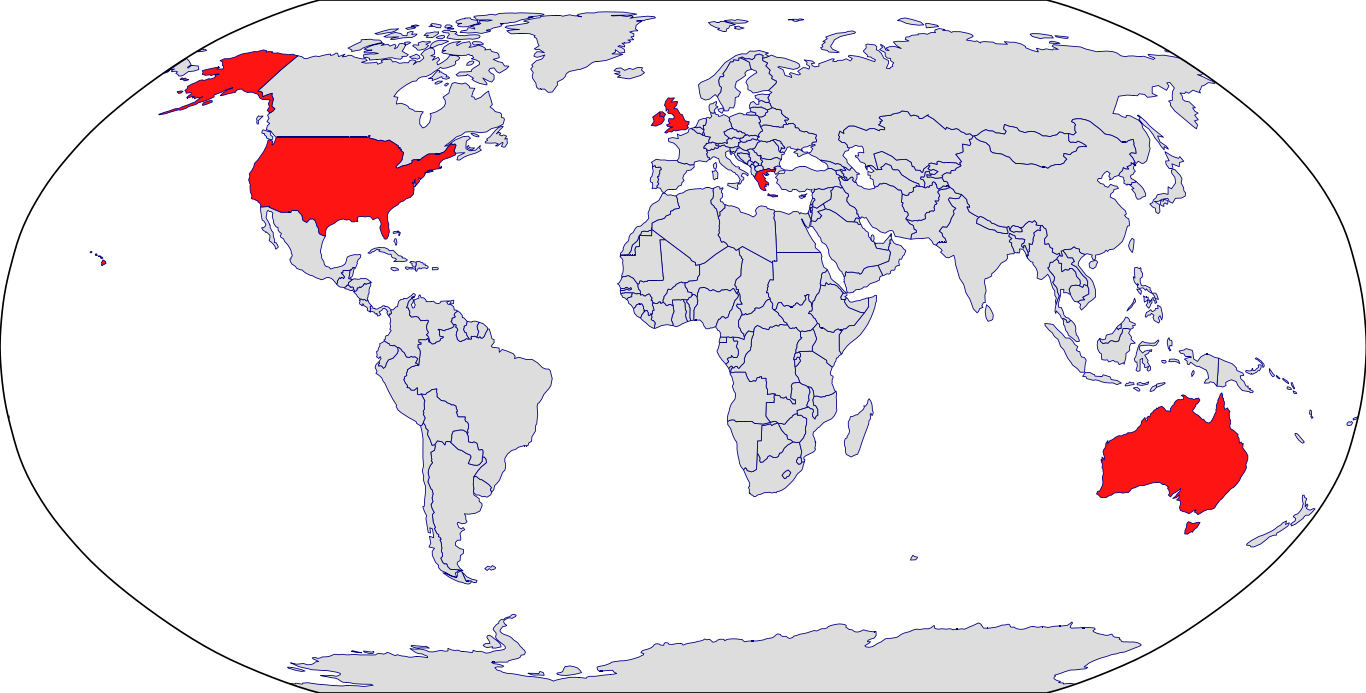
Countries in Clinic: Australia, Greece, Ireland, United Kingdom, United States
Active Clinical Trial Count: 6
Recent & Upcoming Milestones
- Clinical Outcomes Expected - Cybin announced they will present P3 Depressive Disorder, Major results in 4Q26 for Deupsilocin
- Clinical Outcomes Expected - Cybin announced they will present P3 Depressive Disorder, Major results in YE26 for Deupsilocin
- Clinical Outcomes Reported - Cybin presented P2 Depressive Disorder, Major results on 2024-12-10 for Deupsilocin
Highest Development Phases
Phase 3: Depressive Disorder, Major
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT06564818 |
APPROACH | P3 |
Recruiting |
Depressive Disorder, Major |
2026-08-01 |
26% |
2025-09-04 |
Primary Completion Date|Primary Endpoints|Start Date|Study Completion Date|Treatments |
NCT05385783 |
CYB003-001 | P2 |
Completed |
Depressive Disorder, Major |
2023-10-16 |
12% |
2024-03-07 |
|
NCT06605105 |
EXTEND | P3 |
Enrolling by invitation |
Depressive Disorder, Major |
2028-03-15 |
26% |
2026-03-12 |
Primary Endpoints|Treatments|Trial Status |
2024-516805-22-00 |
CYB003-004 | P3 |
Not yet recruiting |
Depressive Disorder, Major |
2027-09-06 |
|||
NCT06793397 |
EMBRACE | P3 |
Recruiting |
Depressive Disorder, Major |
2027-03-18 |
28% |
2025-11-26 |
Primary Endpoints|Start Date|Treatments|Trial Status |
2024-519270-40-00 |
CYB003-003 | P3 |
Not yet recruiting |
Depressive Disorder, Major |
2027-03-18 |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
01/05/2026 |
News Article |
HELUS Pharma Propels Therapeutic Innovation in Mental Health and Commences Trading on Nasdaq |
|
12/30/2025 |
News Article |
Cybin Initiates At-The-Market Equity Program of up to US$100 Million |
|
12/18/2025 |
News Article |
Cybin to Transfer U.S. Stock Exchange Listing to Nasdaq |
|
11/13/2025 |
News Article |
Cybin Reports Second Quarter Fiscal Year 2026 Financial Results and Recent Business Highlights |


