Product Description
Vadadustat is an oral hypoxia-inducible factor (HIF) prolyl hydroxylase inhibitor, a class of drugs that stabilize HIF and stimulate erythropoietin and red-cell production. (Sourced from: https://www.nejm.org/doi/full/10.1056/NEJMoa2035938)
Mechanisms of Action: HIF Inhibitor
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Approved
Approved Countries: Belgium | Croatia | European Medicines Agency | Finland | Iceland | Japan | Korea | Lithuania | Poland | Portugal | Slovakia | Sweden
Approved Indications: None
Known Adverse Events: None
Company: Akebia
Company Location: Eastern America
Company Founding Year: 2012
Additional Commercial Interests: Otsuka
Clinical Description
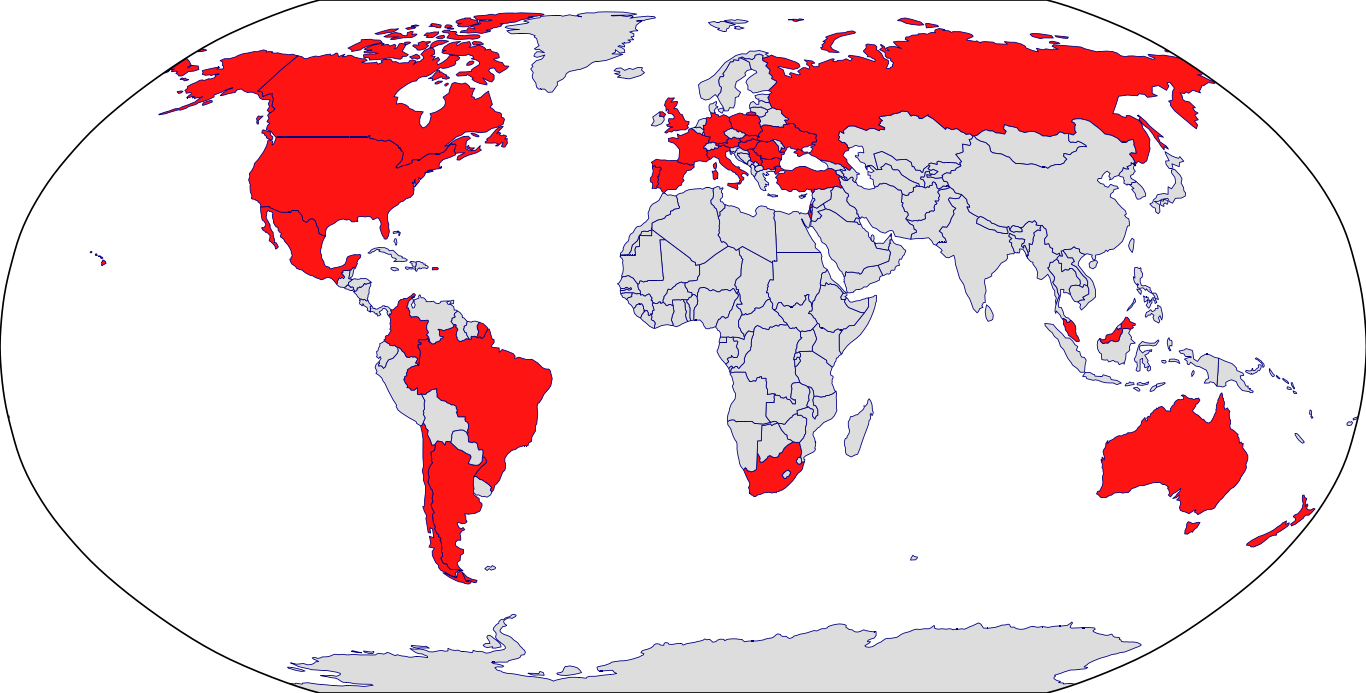
Countries in Clinic: United States
Active Clinical Trial Count: 4
Recent & Upcoming Milestones
- Clinical Outcomes Reported - Akebia presented P3 Kidney Diseases results on 2025-11-06 for Vadadustat
- Clinical Outcomes Reported - Akebia presented P3 Kidney Diseases results on 2025-06-04 for Vadadustat
- Clinical Outcomes Reported - Akebia presented P3 Anemia results on 2025-04-10 for Vadadustat
Highest Development Phases
Phase 3: Acute Lung Injury|Acute Respiratory Distress Syndrome|Anemia|Kidney Failure, Chronic
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT07086755 |
HSC-MS-22-0260 | P3 |
Recruiting |
Acute Respiratory Distress Syndrome|Acute Lung Injury |
2026-07-01 |
2025-11-05 |
Primary Endpoints|Start Date|Treatments|Trial Status |
|
NCT06520826 |
VOICE | P3 |
Active, not recruiting |
Kidney Failure, Chronic|Anemia |
2026-06-30 |
19% |
2026-03-11 |
Primary Endpoints|Treatments|Trial Status |
NCT06901505 |
AKB-6548-CI-0052 | P3 |
Active, not recruiting |
Anemia|Kidney Failure, Chronic |
2026-08-01 |
23% |
2026-02-24 |
Patient Enrollment|Primary Completion Date|Primary Endpoints|Study Completion Date|Treatments|Trial Status |
NCT04707768 |
AKB-6548-CI-0039 | P3 |
Completed |
Kidney Failure, Chronic|Anemia |
2023-01-06 |
30% |
2025-01-16 |
Primary Endpoints |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
01/12/2026 |
News Article |
Akebia Therapeutics Announces Corporate Updates and 2026 Pipeline Outlook |
|
12/01/2025 |
News Article |
Akebia Announces Establishment of Rare Kidney Disease Pipeline |
|
10/28/2025 |
News Article |
Akebia Therapeutics Provides Update on Vafseo for Non-Dialysis Patients |
|
10/20/2025 |
News Article |
Akebia Therapeutics Announces Five Presentations at ASN Kidney Week 2025 |


