Product Description
Pneumococcal Vaccine (Sourced from: https://clinicaltrials.gov/ct2/show/NCT01175083)
Mechanisms of Action: Vaccine
Novel Mechanism: No
Modality: Vaccine
Route of Administration: Intramuscular
FDA Designation: *
Approval Status: Not Approved
Approved Countries: Australia | Austria | Bangladesh | Belgium | Brazil | Chile | Colombia | Croatia | Cyprus | Denmark | Dominican Republic | Egypt | Estonia | Germany | Hungary | India | Indonesia | Italy | Japan | Jordan | Malaysia | Mexico | Morocco | Netherlands | New Zealand | Norway | Pakistan | Peru | Philippines | Poland | Russia | South Africa | Spain | Sri Lanka | Thailand | Tunisia | Turkey | United Arab Emirates | United Kingdom | Venezuela | Vietnam
Approved Indications: None
Known Adverse Events: None
Company: GSK
Company Location: Europe
Company Founding Year: 2000
Additional Commercial Interests: None
Clinical Description
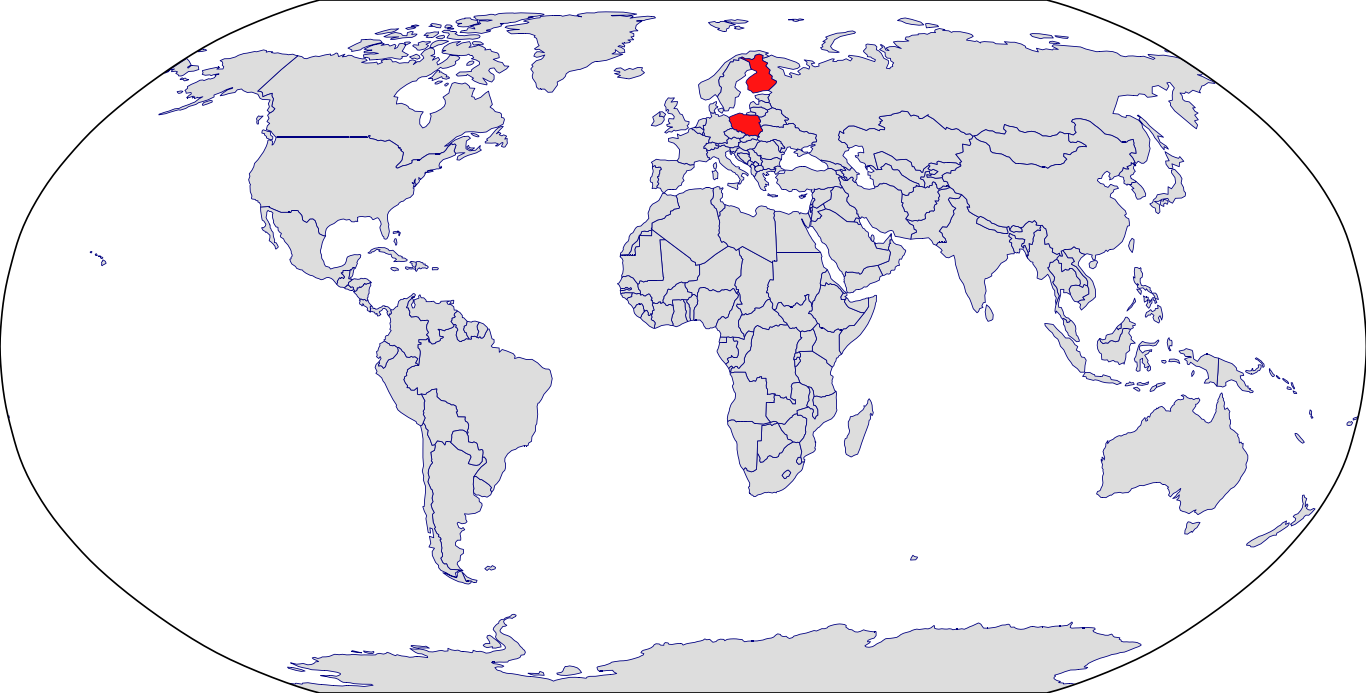
Countries in Clinic: Finland, Poland
Active Clinical Trial Count: 1
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Pneumococcal Infections
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
2023-505154-18-00 |
C4801002 | P2 |
Completed |
Pneumococcal Infections |
2024-05-27 |
2025-05-02 |
Treatments |


