Product Description
VK2809, is a novel, orally available small molecule thyroid hormone receptor agonist that possesses selectivity for liver tissue, as well as the beta receptor sbtype, suggesting promise for the treatment of metabolic disorders, including NASH (Sourced from: https://www.vikingtherapeutics.com/pipeline/metabolic-disease-program/)
Mechanisms of Action: THR Agonist
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Viking
Company Location: Western America
Company Founding Year: None
Additional Commercial Interests: None
Clinical Description
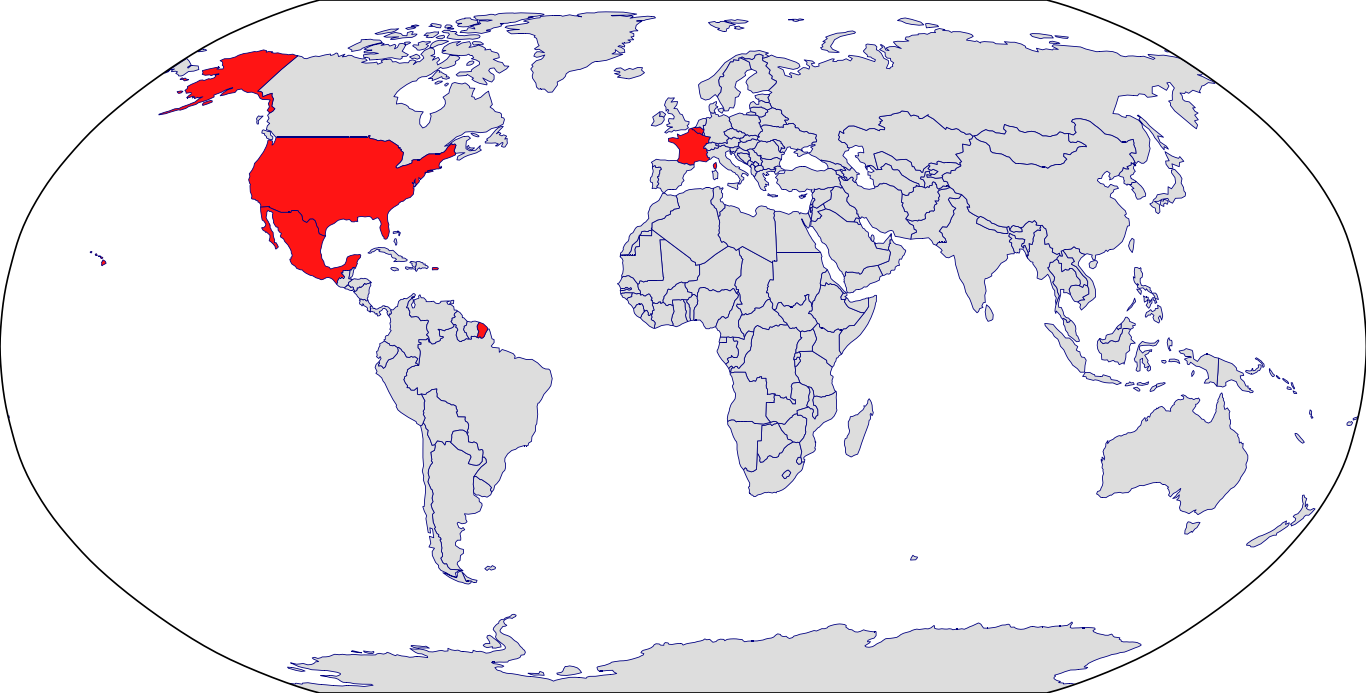
Countries in Clinic: Belgium, France, Mexico, Puerto Rico, United States
Active Clinical Trial Count: 2
Recent & Upcoming Milestones
- Clinical Outcomes Reported - Viking presented P2 Non-alcoholic Steatohepatitis results on 2024-11-19 for VK-2809
- Clinical Outcomes Reported - Viking presented P2 Non-alcoholic Steatohepatitis results on 2024-06-04 for VK-2809
- Clinical Outcomes Reported - Viking announced they will present P2 Non-alcoholic Steatohepatitis results in 2Q24 for VK-2809
Highest Development Phases
Phase 2: Fatty Liver, Alcoholic|Hepatitis, Alcoholic|Non-alcoholic Steatohepatitis
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT04173065 |
VOYAGE | P2 |
Completed |
Fatty Liver, Alcoholic|Non-alcoholic Steatohepatitis|Hepatitis, Alcoholic |
2023-05-15 |
43% |
2024-07-23 |
Primary Endpoints|Study Completion Date|Treatments|Trial Status |
2020-000217-32 |
VOYAGE | P2 |
Active, not recruiting |
Hepatitis, Alcoholic|Non-alcoholic Steatohepatitis |
2022-10-15 |


