Product Description
Mechanisms of Action: Imaging Agent
Novel Mechanism: No
Modality: Diagnostic Agent
Route of Administration: Injection
FDA Designation: *
Approval Status: Not Approved
Approved Countries: None
Approved Indications: None
Known Adverse Events: None
Company: Blue Earth Therapeutics
Company Location: Europe
Company Founding Year: None
Additional Commercial Interests: None
Clinical Description
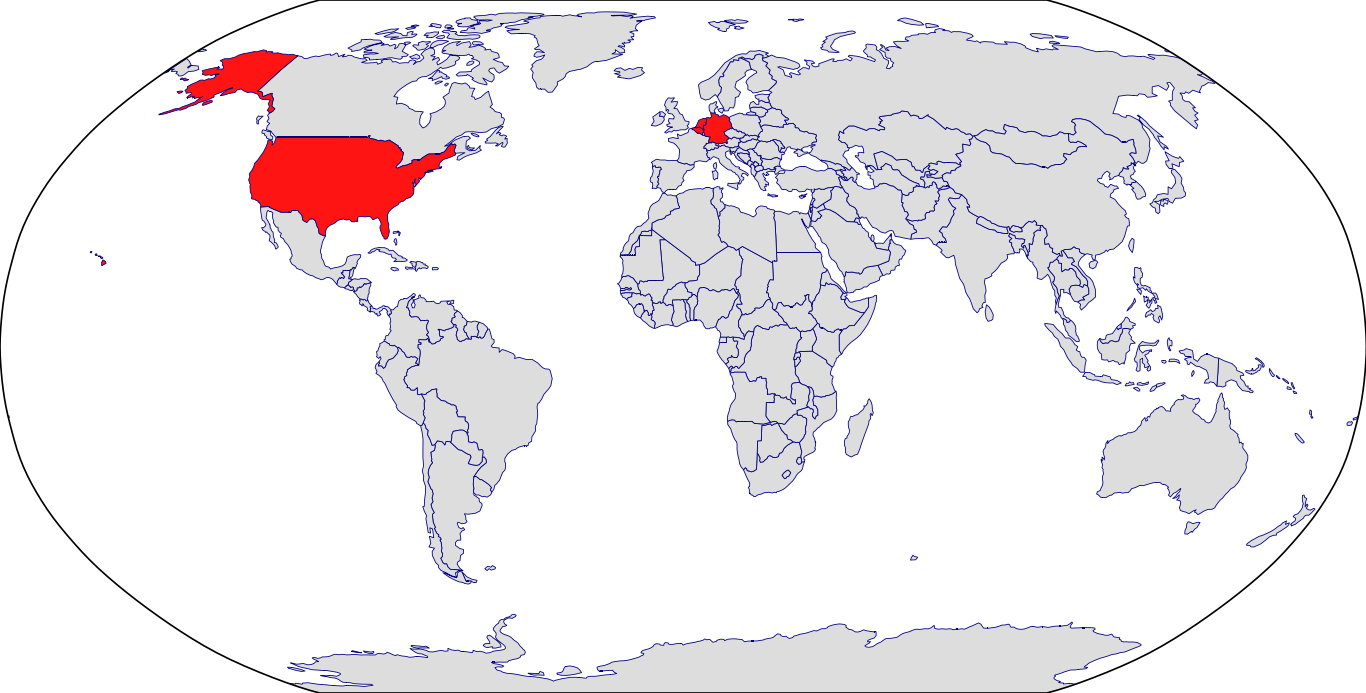
Countries in Clinic: Belgium, Germany, Netherlands, United States
Active Clinical Trial Count: 3
Recent & Upcoming Milestones
Highest Development Phases
Phase 2: Prostate Cancer|Prostatic Diseases|Urogenital Cancer
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05413850 |
BET-PSMA-121 | P2 |
Recruiting |
Urogenital Cancer|Prostate Cancer|Prostatic Diseases |
2026-08-27 |
2% |
2025-03-18 |
Patient Enrollment|Primary Endpoints|Study Completion Date|Treatments |
2022-002407-37 |
2022-002407-37 | P2 |
Active, not recruiting |
Prostate Cancer |
2028-07-05 |
|||
2024-511537-35-00 |
BET-PSMA-121 | P2 |
Recruiting |
Prostate Cancer |
2028-03-31 |
2025-05-02 |
Treatments |


